|
|
|
Indian Pediatr 2017;54: 55-57 |
 |
Chronic Hypersensitivity
Pneumonia due to Pigeon Breeders’ Disease
|
|
Wonashi R Tsanglao, Devki Nandan, Sudha Chandelia and
*Minakshi Bhardwaj
From Departments of Pediatrics and *Pathology,
PGIMER, Dr RML Hospital, New Delhi, India.
Correspondence to: Dr Devki Nandan, Professor,
Department of Pediatrics, PGIMER, Dr RML Hospital, New Delhi 110 001,
India.
Email:
[email protected]
Received: November 24, 2015;
Initial review: March 05, 2016;
Accepted: November 01, 2016.
|
Background: Pigeon breeders’ disease usually affects adults.
Children are more likely to be affected when they share living space
with a backyard poultry or pigeon breeding. Case characteristics:
A 12-year-old girl with persistent cough for 3 years and dyspnea for 2
years. Obervation: She was dignosed to be having allergy to
pigeon droppings, based on reports of lung biopsy and allergy testing.
Message: Pigeon breaders’ disease should be considered in a child
who presents with features of chronic hypersensiticity pneumonitis.
Keywords: Allergy testing, Lung biopsy, Persistent pneumonia.
|
|
P
igeon breeders’ disease (PBD) is one of the most
widespread forms of hypersensitivity pneumonia [1], described
infrequently in children. We describe a girl with chronic
hypersensitivity pneumonia, who presented with prolonged cough and
progressive dyspnea.
Case Report
A 12-year-old girl was referred to us with persistent
dry cough for 3 years, progressive dyspnea for last 2 years and
significant weight loss for last 1 year. She had received
anti-tubercular and anti-asthma treatment without any relief. Her past
and family history was insignificant, except that they were involved in
breeding around 60 pigeons at home. A recent chest X-ray revealed
bilateral ground glass haziness and pulmonary function tests (PFT)
revealed severe restriction (FVC 16.6 %, FEV1 17.9 % , FEV1/FVC 1.07 ),
with reduced diffusion capacity of lung for carbon monoxide (DLCO)
(19%).
On examination, she was cachexic and dyspneic at
rest, with tachycardia and tachypnea. Her S PO2
was 82% at room air that improved to 95% with supplemental oxygen.
Respiratory system examination revealed use of accessory muscles of
respiration, with pectus excavatum and fine basal crepitations.
Cardiovascular system examination revealed loud P2, and normal jugular
venous pressure and hepato jugular reflux. She had no hepatomegaly. She
weighed 23 kgs (< -3 SD), with a height of 144 cm (-2 to -3 SD) and body
mass index of 11.11 (< -3SD). She was in tanner stage II of sexual
maturity.
On investigation, arterial blood gas revealed PO 2
of 70 mmHg, with saturation of 91%. Echocardiography revealed dilated
pulmonary artery, dilated right ventricle and moderate tricuspid
regurgitation (gradient 30 mm of Hg) suggestive of pulmonary artery
hypertension (PAH). Work up for tuberculosis, immunodeficiency (primary
and secondary), sarcoidosis, connective tissue disorders, celiac
disease, tropical pulmonary eosinophilia, allergic bronchopulmonary
aspergillosis, cytomegalovirus and mycoplasma were negative. Fibreoptic
bronchoscopy was grossly normal. Cytology of bronchoalveolar lavage and
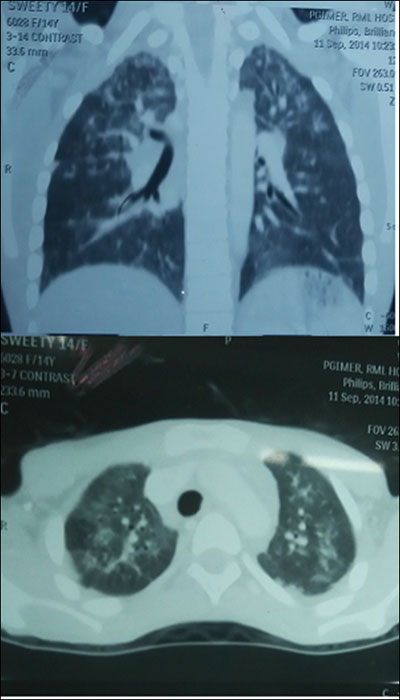
flow cytometry were inconclusive. High resolution computed tomography
(HRCT) of chest (Fig. 1) revealed diffuse mosaic pattern
and multiple ill-defined centrilobular nodular lesions in both upper
lobes and interstitial thickening in the apical segment of left lower
lobe. Histopathology of lung biopsy specimen revealed classical features
of hypersensitivity Pneumonia (Web Fig.1) [2,3]. Test for
pigeon dropping allergy in serum was positive.
 |
|
Fig. 1 Patchy areas of ground glass
haze seen in both upper lobe with air trapping (white arrow)
giving rise to mosaic pattern and multiple ill-defined
centrilobular nodular lesions in both upper lobes (black arrows)
and interstitial thickening in the apical segment of left lower
lobe.
|
She was administered intravenous methylpre-dnisolone
(30 mg/kg/day) for 3 days, followed by oral prednisolone and inhaled
budesonide. Bosentan (BOSENTAS 62.5 mg twice daily) [4] was also added
in view of the pulmonary hypertension. At day 45, she was discharged on
home oxygen, and advised to avoid exposure to pigeons. Her weight at
discharge was 24 kg. She was having dyspnea only on exertion, with SpO 2
of 92% at room air and FEV1 and FVC of 29% and 23%, respectively. Oral
steroids and bosentan were continued. Diuretics were not administered as
she had no features of congestive heart failure. She was followed-up
with monthly monitoring of respiratory rate, SpO2,
pulmonary function and liver function test. Bosentan was stopped after
two-dimentional echocardiography at eight weeks showed no evidence of
pulmonary hypertension. She was continued on low dose prednisolone. At
10 months follow-up, she was on room air, attending school; FEV1 and FVC
were 33% and 37%, respectively.
Discussion
Hypersensitivity pneumonia is an immune-mediated lung
parenchymal injury occurring in response to repeated inhalation of an
antigen. Antigens implicated include animal proteins, fungi, amoeba,
bacteria, medications and chemicals. Avian antigens are one of the most
common causes of hypersensiticvity pneumonia [2]. Both type III and type
IV hypersensitivity responses have been implicated in the disease
process [5]. The chronic form of hypersensitivity pneumonia results from
long term low-grade exposure, and is characterized by dyspnea, chronic
cough, fatigue, anorexia and weight loss. PFT typically reveals a
restrictive pattern and a decrease in DLCO. HRCT chest in chronic
hypersensiticvity pneumonia reveals fibrotic changes, irregular linear
opacities, centri-lobular nodules, honeycombing and traction
bronchiectasis [2]. Bronchoalveolar lavage usually reveals a significant
increase in the percentage of lymphocytes with decreased CD4+/CD8+ratio
[6], On histopathology, the interstitial and alveolar collections of
foamy histiocytes are considered to be fairly specific for
hypersensitivity pneumonia due to pigeon breeder’s disease [3], which
was observed in our case. Interstitial fibrosis and interstitial
cellular infiltrates that is primarily lymphocytic with large number of
plasma cells, with absence of granulomas is also observed in chronic
hypersensitivity pneumonia.
Common differentials include other interstitial lung
diseases which include immune-mediated collagen vascular diseases,
sarcoidosis, langerhans cell histiocytosis and malignancies [7-8].
Diagnostic criteria for hypersensitivity pneumonia include six major and
three minor criteria [9]. Establishing diagnosis requires the presence
of at least 4 major and 2 minor criteria. The present case had 5 major,
and all 3 minor criteria. Treatment for chronic hypersensitivity
pneumonia includes oral prednisolone over several months, depending on
the response to improvement in symptoms and functional abnormalities
[10].
Early treatment leads to complete reversal in acute
and sub-acute hypersensitivity pneumonia. Chronic form may proceed to
irreversible lung damage in spite of treatment and avoidance of the
offending antigen. Our case continues to have restrictive changes at 10
months follow-up. The clinicians should have a high index of suspicion
in order to make early diagnosis and avoid disease progression and
irreversible lung damage.
Contributors: DN: supervised the diagnosis and
management of the case and revised the manuscript; WRT: searched the
literature and drafted the manuscript;; SC: searched the literature and
critically reviewed the manuscript. MB: tissue diagnosis and searched
the literature All authors approved the final manuscript.
Funding: None; Competing interest: None
stated.
References
1. Grech V, Vella C, Lenicker H. Pigeon breeder’s
lung in childhood: Varied clinical picture at presentation. Pediatr
Pulmonol. 2000;30:145-8.
2. Kurup VP, Zacharisen MC, Fink JN. Hypersensitivity
pneumonitis. Indian J chest Dis Allied Sci. 2006;48: 115-28.
3. Riley DJ, Saldana M. Pigeon breeder’s lung.
Subacute course and the importance of indirect exposure. Am Rev Respir
Dis. 1973;107:456-60.
4. Abman SH, Hansmann G, Archer SL, Ivy DD, Adatia I,
Chung WK, et al. Pediatric Pulmonary Hypertension Guidelines from
the American Heart Association and American Thoracic Society.
Circulation. 2015;132:2037-99.
5. Du Marchie Sarvaas GJ, Merkus PJ, De Jongste JC. A
family with extrinsic allergic alveolitis caused by wild city pigeons: a
case report. Pediatrics. 2000;105:e62.
6. Ratjen F, Costabel U, Griese M, Paul K.
Bronchoalveolar lavage fluid findings in children with hypersensitivity
pneumonitis. Eur Respir J. 2003;21:144-8.
7. Fan LL, Deterding RR, Langston C. Pediatric
interstitial lung disease revisited. Pediatr Pulmonol. 2004;38:369-78.
8. Deutsch GH, Young LR, Deterding RR, Fan LL, Dell
SD, Bean JA, et al. Diffuse lung disease in young children:
application of a novel classification scheme. Am J Respir Crit Care Med.
2007;176:1120-8.
9. Schuyler M, Cormier Y. The diagnosis of
hypersensitivity pneumonitis. Chest. 1997;111:534-6.
10. Fink JN, Zacharisen MC. Hypersensitivity
Pneumonitis. In: Adkinson NF, Yunginer JW, Busse WW, Bochner BS,
Holgate ST, Simons FE. Middleton's Allergy: Principles and Practice. 6th
ed. Philadelphia: Mosby and Co; 2003. p. 1373-90.
|
|
|
 |
|

