|
|
|
Indian Pediatr 2013;50:
203-207 |
 |
Expressed Breast Milk vs 25% Dextrose
in Procedural Pain in Neonates: A Double Blind Randomized
Controlled Trial
|
|
Jagdish Prasad Sahoo, Suman Rao, Saudamini Nesargi, Thomas Ranjit, Ashok
C and Swarnarekha Bhat
From Department of Neonatology, St John’s Medical College and
Hospital, Bangalore, Karnataka, India.
Correspondence to: Dr Jagdish Prasad Sahoo, Senior Resident,
Department of Pediatrics, Kalinga Institute of Medical Sciences,
Bhubaneswar, Orissa, India.
Email:
[email protected]
Received: December 09, 2011;
Initial review: January 06, 2012:
Accepted: June 23, 2012.
Published online: 2012, July 5.
PII: S097475991101008-1
|
Objective : To
compare the effect of expressed breast milk (EBM), 25% dextrose (25 D)
and sterile water (SW) on procedural pain in neonates as assessed by the
premature infant pain profile (PIPP), changes in heart rate (HR), oxygen
saturation (SpO2) and duration of crying.
Design: Prospective, double blind, randomized
controlled trial.
Setting: Postnatal ward of a tertiary-care
hospital.
Participants: 210 babies who required
venipuncture for blood sampling and who were on oral feeds were
recruited into the study after parental informed consent.
Methods: The enrolled babies were randomized into
intervention groups (EBM, 25% dextrose) and control group (sterile
water). Two ml of test solution was given to baby by paladay (a
traditional cup with a spout) 2 min before venipuncture. The face and
crying of baby were video graphed by an independent, blinded observer.
The facial response to pain (brow bulge, eye squeeze, nasolabial furrow)
was analysed from the video. Maximum HR and minimum SpO2 were recorded
during, and 1, 3 and 5 min after venipuncture by another blinded
observer.
Outcome variable: :PIPP score, HR, SpO2 and
crying time at 0/1/3/5 min after sampling.
Results: 160 babies were considered for final
analysis with 50 in 25 D, 62 in EBM and 48 in SW group. The mean PIPP
score in the 3 groups were 5.22, 6.84 and 11.22 at 0-30 sec after
venipuncture; 4.52, 6.34, and 10.88 at 1-1 ½ min; 3.96, 6.15 and 9.35 at
3-3 ½ min; and 3.12, 4.68 and 7.83 at 5-5 ½ min; respectively (P<
0.001). The median crying time was 10 ,37.5 and 162 seconds in 25
D, EBM and SW groups, respectively (P< 0.001).
Conclusions: EBM significantly reduces procedural
pain in neonates though to a lesser extent as compared to 25% dextrose.
Key words: 25% dextrose, Expressed breast milk, Neonates,
Procedural pain, PIPP score.
|
|
Most newborns routinely undergo painful invasive
procedures in the hospital. It is now well recognized that even preterm
neonates are anatomically and physiologically capable of feeling pain.
Pain in neonates can evoke negative behavioral, physiologic, or
metabolic responses [1], and
may be associated with long term consequences. As neonates cannot
verbalize their pain, they depend on others to recognize, assess and
manage their pain.
Many pharmacologic and non-pharmacologic methods have
been proposed for pain control in neonates [2-5]. Despite these, the
pain during various neonatal procedures is inadequately controlled.
Expressed breast milk [6,7], sucrose [7,8]
and glucose (10-33%) [9,10] have been used in various
trials for reducing procedural pain in neonates with equivocal effects.
We hypothesized that both expressed breast milk and 25% dextrose are
equally effective in reducing pain from venipuncture. We tried to
compare the efficacy of expressed breast milk and 25% dextrose in
comparison with sterile water in reducing pain during venipuncture in
neonates as assessed by the Premature Infant Pain Profile.
Methods
This prospective randomized controlled trial was
conducted in the post-natal ward of a tertiary care hospital in southern
India from April 2010 to September 2010. Babies
³34 week gestations
that required venipuncture for blood sampling and who were on oral feeds
were included in the study after informed parental consent. Neonates who
were sick, had perinatal asphyxia (Apgar score <5 at 5 minutes),
congenital malformations or were on opioid analgesics, sedatives or
phenobarbi-tone were excluded from the study. After inclusion, all
neonates who cried before venipuncture or passed stool/urine during
sampling or had more than one prick were excluded from analysis, as
decided a priori. The study was cleared by the Institutional
ethical review board.
The eligible babies were randomized into 3 groups:
two intervention groups - Expressed breast milk group (EBM), 25%
dextrose group (25D) and sterile water group (SW) – the control group;
using computer generated random numbers. Allocation concealment was
achieved by using sequentially numbered opaque sealed envelope
containing the codes for intervention (EBM for expressed breast milk,
25D for 25% dextrose, SW for sterile water). The envelopes were
exclusively accessed by the principal investigator.
The neonates requiring venipuncture were taken to a
quiet room. It was ensured that time interval between the procedure and
previous breast milk intake was at least one hour. The babies’
gestational age was determined by New Ballard Score and behavioral state
was recorded. Oxygen saturation (SpO 2)
and heart rate (HR) were monitored by a pulse oxymeter (MASIMO). Two ml
of test solution was administered to the baby through a sterile
paladay (a traditional cup with a spout) by mouth by one staff
nurse. The excess amount of test solution and the paladay were
cleared before the entry of observers into the room. All venipunctures
were done with 23 gauge needle 2 minutes after the test solution was
administered. All venipunctures were done by one of a selected group of
staff nurses who had adequate experience in neonatal care to minimize
variation in pain during venipuncture. Two observers were present during
sampling time. One observer videographed the face of baby for later
analysis; the other observer was responsible for recording the HR, SpO2
and duration of venipuncture. The observers entered the room after test
solution was administered, and thus were masked to the test solution
given. HR and SpO2 were
recorded at baseline, and at 0, 1, 3, 5 min after venipuncture. Maximum
HR and minimum SpO2 were
noted between 0-30 seconds, 1-1½ min, 3-3½ min and 5-5½ min after the
venipuncture. Crying time was defined as the total duration of audible
cry which was recorded from the video recording. Sampling time was
defined as the time gap between the times of insertion of needle for
venipuncture to the time the needle was removed from the baby. The
facial response to pain (brow bulge, eye squeeze, nasolabial furrow) in
this period was recorded and subsequently analyzed from the video by a
single observer. The primary outcome of the study was the PIPP score
[11]. The PIPP score is a composite pain measure that includes
contextual (behavioral state and gestational age), behavioral (brow
bulging, eye squeezing and nasolabial furrowing), and physiologic (heart
rate and oxygen saturation) indicators of pain. Each indicator is scored
in a 4 point scale (0-3) and pain intensity scores range from 0-21.
Scores of 6 or less represent absence of pain or minimal pain. PIPP
score was not calculated in case of difficulty in analyzing the facial
response from the videos. The secondary outcomes were change in HR, SpO2
and crying time.
The sample size was calculated based on the study by
Taddio, et al. [8], where the mean difference of PIPP score was
1.8 between the two study groups (24% sucrose and placebo) group.
Considering the standard deviation for the group 1 as 2.9 and standard
deviation for the group 2 as 2.5, with 1% level of significance and 90%
power, 201 subjects were to be recruited, 67 subjects in each arm
(breast milk, 25% dextrose, sterile water). However the sample was
increased to 210 (70 in each group) to account for the possible loss of
data.
Statistical analysis was done with ANOVA (analysis of
variance) to assess the effect within and between the groups. Analysis
was done with SPSS statistical software package 16. Post-hoc analysis
was done using Bonferroni test. Crying time was analyzed by Kruskal
Wallis test. Repeated measures ANOVA were used to compare the heart rate
and SpO 2 over time and it’s
interaction with the intervention group. A P value of 0.01 was
considered a statistically significant level of difference.
Results
A total of 210 babies were randomly allocated to the
3 groups (25 D, EBM, and SW). 50 neonates were excluded from the
analysis because of incomplete data (Fig 1). The analysis
included 160 babies (50 in 25 D, 62 in EBM and 48 in SW group). No
differences were observed between the groups with regard to baseline
variables (Table I).
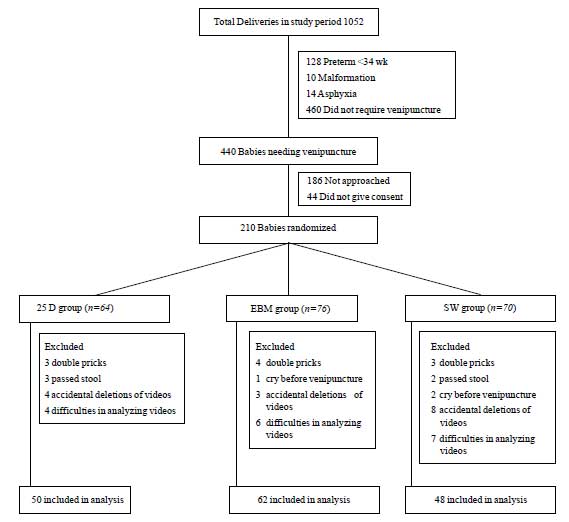 |
|
Fig. 1 Trial profile and participant
flow.
|
TABLE I Baseline Characteristics of Study Population
|
Parameter |
25D group (n=50) |
EBM group (n=62) |
SW group (n=48) |
|
Birth weight* (g) |
2965 (2507-3287) |
2605 (2226-2992) |
2720 (2297-3000) |
|
Gestation# (wk) |
37.94 ( 1.64) |
37.66 (1.88) |
37.93 (1.61) |
|
Male |
30 (60%) |
44 (70%) |
28 (58%) |
|
Vaginal delivery |
25 (50%) |
35 (56%) |
23 (48%) |
|
Postnatal age (hr)* |
77 (55.5-102) |
84 (61.5-97.5) |
89 (54-98) |
|
Time since last feed# (min) |
59.7 (8.1) |
56.84 (7.4) |
58.54 (6.6) |
|
Sampling time* (sec) |
50 (38.5-60) |
51.5 (41.2-63.7) |
55 (43.7-65) |
|
*median (IQR); #Mean (SD); EBM – Expressed breast milk; SW –
Sterile Water; 25 D – 25% dextrose. |
The crying time [median (IQR)] was lower in the 25%
dextrose group [10(0-70)] as compared with the EBM [37.5 (5-142)] and
sterile water group [162 (59-200)]. The difference between crying time
in 25D and EBM group was not significant. A significant difference was
found between SW group compared to 25D and EBM groups.
There was a significant change in HR and SpO 2
over time (P<0.001). The HR and SpO2
observed in the sterile water group were significantly higher and lower,
respectively when compared to the other two groups for all time points
except baseline (Table II).
TABLE II Heart Rate and SPO2 [Mean (SD)] in The Study Subjects
|
|
Heart rate (beats/min)
|
SPO2 (%) |
|
25D |
EBM |
SW |
25 D |
EBM |
SW |
|
Baseline |
131.7 (13.9) |
127.4 (16.2) |
125.3 (14.7) |
97.5 (1.9) |
97.9(1.8) |
98.2 (1.4) |
|
After venipuncture |
|
0 - 30 sec |
142.2 (13.6) |
142.5 (17.4) |
153.8 (15.5) |
95.2 (3.6) |
95.8(2.1) |
95.1 (1.9) |
|
1 min- 1 ½ min |
140.0 (14.9) |
142.5 (21.4) |
156.4 (19.1) |
95.5 (2.7) |
95.4(3.1) |
93.7 (2.4) |
|
3 min- 3½min |
138.4 (17.0) |
142.0 (21.3) |
155.2 (18.9) |
95.7 (2.3) |
95.7(2.9) |
93.9 (2.5) |
|
5 min-5 ½ min |
132.6 (17.0) |
137.6 (20.9) |
149.4 (17.4) |
96.0 (2.2) |
96.52(2.4) |
94.4 (2.4) |
|
EBM – Expressed breast milk; SW – Sterile Water; 25 D – 25%
dextrose.
|
The mean PIPP score was significantly lower at 0-30
sec, 1- 1 ½ min, 3-3 ½ min, and 5- 5 ½ minutes after the procedure in
the 25 D, and EBM groups vs SW group. Further, the PIPP score was
higher in EBM group than 25 D group at all time points.
The PIPP score was not significantly different at 30 sec between the 25D
and EBM group (P=0.58) (Table III).
TABLE III PIPP Score in the Study Subjects
|
25 D group* |
EBM group |
SW group |
|
0 - 30 sec |
5.22 (4.3-6.1) |
6.84 (5.8-7.8) |
11.21 (10.1-12.2) |
|
1 min- 1½ min |
4.52 (3.6-5.4) |
6.34 (5.2-7.4) |
10.88 (9.7-12) |
|
3 min- 3½ min |
3.96 (3.2-4.6) |
6.15 (5.1-7.1) |
9.35 (8.2-10.4) |
|
5 min-5½ min |
3.12 (2.4-3.8) |
4.68 (3.8-5.5) |
7.83 (6.7-8.8) |
|
* PIPP score in 25 D vs. EBM group at 1- 1 ½min, 3- 3 ½ min,
5- 5 ½ min P=0.042, P= 0.003, P=0.036, respectively. |
There were no adverse events in any of the groups
except for 1 baby having transient bradycardia (HR <100) in the 25D
group.
Discussion
This study showed that both 25% dextrose and breast
milk decrease pain response (behavioral and physiologic) in newborn
babies as assessed by the PIPP score. The mean crying time, heart rate
and oxygen saturation changes at 0, 1, 3, 5 minute after venipuncture
were significantly reduced in the 25% dextrose or breast milk group as
compared to placebo (sterile water), and the analgesic effect persisted
till 5 minutes after the procedure. The analgesic effect of 25% dextrose
or expressed breast milk is probably based on the link that exists
between the oro-gustatory effects of sweet solution given orally and the
endogenous opioid pathway [12, 13].
In all probability this is due to the sweet taste
perception, a sense well developed even in premature infants at birth.
Though oral sucrose has been widely studied as an
analgesic tool and has been found to be safe and effective [14], we
preferred to use 25% dextrose, as sucrose is not readily available in
the neonatal units in India. Compared to sucrose, glucose is 0.75 times
as sweet [15] and 25% dextrose is commercially available as sterile
ampoules. Breast milk also has analgesic properties and has been found
to reduce pain from procedures. The analgesic effect of breast milk may
be related to the sweetness of breast milk (presence of lactose in
breast milk) [16] or higher concentration of tryptophan, a precursor of
melatonin that increase the concentration of beta endorphins [17]. Being
a natural food, it would be the most ideal and safe analgesic. Also it
is readily available, easy to use and can be repeated without risk.
Similar analgesic effects of expressed breast milk
were observed in studies by Upadhyaya, et al. [6]. They had;
however, used a higher volume (5 mL) of EBM. Others found a better
response with higher glucose concentration (25-30%) compared to 10%
glucose or breast milk [9,10]. In contrast to our study, Desmukh, et
al. [10] did not find significant reduction in heart rate and oxygen
saturation in their study on preterm infants. Ors, et al. [7]
also concluded that the antinociceptive effect of human milk is not as
effective as an analgesic as 25% sucrose solution.
We observed that though EBM and 25% dextrose reduced
the mean PIPP score after venipuncture, 25% dextrose was better.
Mariano, et al. [18] compared the efficacy of expressed breast
milk vs 25% glucose on pain responses of late preterm infants
during heel lancing. They also observed lower incidence of cry and
shorter duration of crying in the 25% dextrose group. They concluded
that the results based on PIPP score and crying time indicate better
effects of 25% glucose compared to EBM during heel lancing.
One of the strength of the study is that we have used
a very objective, validated and reliable tool for assessment of pain
response - the PIPP tool [19]. The PIPP adjusts for gestational age and
is also validated for use in term neonates [20]. The other strength of
the study is that the observers were masked to the analgesic used. The
main limitation of our study is the post-randomization exclusions,
because of video errors (difficulty in analyzing/accidental deletion of
some videos). We have neither studied the long term effect of painful
stimuli nor the effect of multiple punctures.
This study further emphasizes that venipuncture is a
painful procedure with pain scores as high as 11 immediately after the
procedure and high scores persisting after 5 minutes; though we did not
record the median recovery time. The use of analgesics, either glucose
or breast milk, does not totally alleviate the pain. Pain management in
newborns must thus be multi- pronged.
Pain in newborn should be recognized and treated. We
conclude that 25% dextrose and expressed breast milk cause effective
reduction in pain response in newborn babies during venipuncture and
there is a better response with 25% dextrose. EBM which is easily
available and useful for alleviating pain response does not totally
alleviate pain and further studies should evaluate groups of
intervention for pain reduction.
Contributors: JPS: conceiving and designing the
study, protocol development, patient screening and analysis of video for
PIPP score, outcome assessment, and writing of manuscript; SR: study
design, writing of manuscript; SN: randomization of study population and
statistical analysis; TR & AC: patient screening, collection of data,
video recording; and SB: supervised the design and execution of study,
outcome assessment, writing of manuscript. She will act as guarantor of
the study. The final manuscript was approved by all the authors.
Funding: None; Competing interests: None
stated.
|
What is Already Known?
• Expressed breast milk (EBM) and 25%
dextrose have pain relieving property.
What This Study Adds?
• EBM significantly reduces procedural pain in neonates,
though to a lesser extent as compared to 25% dextrose.
|
References
1. Anand K J, Carr DB. The neuroanatomy,
neurophysiology and neurochemistry of pain, stress and analgesia in
newborns and children. Pediatr Clin North Am. 1989; 36: 795-822.
2. Carbajal R, Chauvet X, Couderc S,
Olivier-Martin M. Randomized trial of analgesic effects of sucrose,
glucose and pacifiers in term neonates. BMJ. 1999; 319: 1393-7.
3. Gray L, Watt L, Blass EM. Skin-to-skin contact
is analgesic in healthy newborns. Pediatrics. 2000; 105: e14.
4. Corbo MG, Mansi G, Stagni A, Romano A, van den
Heuvel J, Capasso L, et al. Non-nutritive sucking during heel
stick procedures decreases behavioural distress in the newborn
infant. Biol Neonate. 2000; 77:162-7.
5. Carabajal R, Veerapen S, Couderc S, Jugie M,
Ville Y. Analgesic effect of breast feeding in term neonates: a
randomized controlled trial. BMJ. 2003; 326:13-17.
6. Upadhyay A, Aggarwal R, Narayan S, Joshi M,
Paul VK, Deorari AK. Analgesic effect of expressed breast milk in
procedural pain in term neonates: a randomized, placebo-controlled
double blind trial. Acta Pediatr. 2004; 93:518-22.
7. Ors R, Ozek E, Baysoy G, Cebeci D, Bilgen H,
Türküner M, et al. Comparison of sucrose and human milk in
pain response in neonates. Eur J Pediatr. 1999; 158:63-6.
8. Taddio A, Shah V, Hancock R, Smith RW,
Stephens D, Atenafu E, et al. Effectiveness of sucrose
analgesia in newborns undergoing painful medical procedures. CMAJ.
2008; 179:37-43.
9. Skogsdal Y, Eriksson M, Shollin J. Analgesia
in newborns given oral glucose. Acta Pediatr. 1997; 86:217-20.
10. Desmukh LS, Udani RH. Analgesic effect of
oral glucose in preterm infants during venipuncture - a double blind
randomised controlled trial. J Trop Pediatr. 2002; 48:138-41.
11. Stevens B, Johnson C, Petryshen P, Taddio A.
Premature Infant Pain Profile: development and initial validation.
Clin J Pain. 1996; 12:13–22.
12. Blass E, Fitzgerald E, Kehoe P. Interactions
between sucrose, pain and isolation distress. Pharmacol Biochem
Behav. 1987; 26:483-9.
13. Gibbins S, Stevens B. Mechanisms of sucrose
and non-nutritive sucking in procedural pain management in infants.
Pain Res Manag. 2001; 6:21-8.
14. Stevens B, Yamada J, Ohlsson A. Sucrose for
analgesia in newborn infants undergoing painful procedures. Cochrane
Database Syst Rev. 2010 Jan 20; 1:CD001069.
15. Matrindale W. The Extra Pharmacoepia. 29th
edn, London: Pharmaceutical press; 1989: 1275.
16. Blass EM. Milk-induced hypoalgesia in human
newborns. Pediatrics. 1997; 99: 825–9.
17. Barrett T, Kent S, Voudouris N. Does
melatonin modulate beta-endorphin, corticosterone and pain
threshold? Life Sci. 2000; 66:467-76.
18. Buenno M, Stevens B, Camargo P, Toma E, Krebs
V, Kimura A. Breast milk and glucose in pain relief in preterm
infants: A noninferiority randomized controlled trial. Pediatrics.
2012; 129: 2011-2024.
19. Stevens B, Johnson C, Taddio A, Gibbins S,
Yamada J. The premature infant pain profile: evaluation 13 years
after development. Clin J Pain. 2010; 26:813-830.
20. Ballanntyne M, Stevens B, McAllister M, Dionne K, Jack A.
Validation of the premature infant pain profile in the clinical setting.
Clin J Pain. 1999;15: 297-303.
|
|
|
 |
|

