|
The current threat to
tuberculosis (TB) control is due to the emergence of strains
resistant to the two most potent anti-TB drugs viz.,
isoniazid (H) and rifampicin (R) (multidrug
resistant-tuberculosis, MDR-TB). The response of patients with
MDR-TB to treatment is poor and the mortality rate is usually
high [1,2], and situation is further threatened by the emergence
and spread of HIV [1]. Information on drug-resistant (DR) TB is
limited to a small number of patients or is incomplete,
especially in children [1-4]. Thus, this study was done to
define the clinical profile of drug resistant TB in children.
Methods
This cross-sectional observational study was
done at our Pediatric TB Clinic over a period of three years
from the July 2007 to August 2010 after approval from the
institutional ethics committee and informed consent from the
parents of children. Five hundred children less than 15 years of
age who were diagnosed to have TB were studied. Children were
defined to have drug resistant -TB if their drug susceptibility
testing (DST) detected resistance to any of the antitubercular
drugs, or these children were in contact with an adult suffering
from culture proven drug resistance. Specimens used for cultures
were sputum/ bronchoalveolar lavage (BAL) in case of pulmonary
TB, lymph nodes in case of TB lymphadenopathy, pus in case of
bone TB, cerebrospinal fluid (CSF) in case of TB meningitis,
pleural fluid in case of pleural effusion, and DST from contacts
with DR-TB. Children who were clinically suspected to DR- TB but
were not culture proven were excluded from the study. Patients
were classified to have monoresistant TB, polyresistant TB, MDR
TB and XDR-TB as per WHO classification [3]. Others were
classified as contact with DR-TB, and Partial XDR TB if they had
MDR TB along with either a fluoroquinolone or an aminoglycoside
resistance (apart from streptomycin) [4]. Antitubercular
therapy (ATT) susceptibility pattern testing for all 13 drugs
including isoniazid(H), rifampicin(R), pyrazinamide(Z),
ethambutol(E) and streptomycin(S), amikacin (Amk), kanamycin
(Km), fluoroquinolones, capreomycin, Clofazimine (Cfz),
ethionamide (Eth), PAS were done for these patients after
culture on broth and solid media was positive and appropriate
second line drugs were started on them.
A detailed clinical history and physical
examination were done in all patients. History of BCG
vaccination, past TB or contact with TB (contact with a person
who is taking ATT or has taken ATT in past 2 years was elicited.
All patients were screened with chest X-ray and mantoux
test (MT). In case of positive MTB on smear or culture, or a
patient who had a positive MT in the past, MT was not done. A
positive Mantoux test was considered if it was more than 10 mm
with maximum 5 TU units. Latent TB was defined as an
asymptomatic MT positive child in close contact with an adult
having open TB in the past 2 years. Radiological evaluation of
disease progression in form of Chest X-ray, ultrasound of
abdomen and neuroimaging (CT scan and MRI) were done and as and
when required. Tissue cultures for repeat DST testing were done
when feasible. Pre-test HIV counselling was done in all patients
as per WHO criteria [5] and HIV-ELISA was done only after
consent in these patients. Investigations such as hemogram,
liver and renal biochemistry, uric acid, thyroid function tests
and ophthalmological examination for colour blindness along with
hearing assessment were done at start of therapy as well as
bimonthly intervals when these patients were put on second line
ATT.
Clinical and biochemical features associated
with drug resistant-TB were analysed. The children were divided
into 3 age groups viz. <5 years, 5-10 years, and >10
years, and factors associated with drug resistant TB in various
age groups both sexes were analyzed. Since patients are on
therapy at time of study, their response to treatment has not
been assessed in this study. Statistical significance between
the mean was calculated by student t test and analysis of
variance (ANOVA). Proportions were analysed by Fisher Exact
test. P<0.05 was considered significant.
Results
During the study period, drug-resistant TB
was seen in 34 (6.8%) children (Fig. 1). The
specimens used was from sputum/BAL in 17 (50%) children, lymph
nodes in 8 (23.5%) children, CSF in 2 (5.8%) and pus and pleural
fluid in 1 (2.9%) patient each. Five (14.7%) children were
defined as DR-TB based on DST report of the contacts. Mean age
of presentation of DR-TB was 6.8 ± 3.2 years. Male: Female ratio
was 13:21. Common clinical features of DR-TB and their duration
are depicted in Web Table I. Eighteen children
(52.9%) had been treated for tuberculosis in the past and took
ATT for 10.6 months of which 1 (5.5%) defaulted. The type of TB
that they had suffered from in the past were abdominal TB in 3
(16.7%), pulmonary TB in 12 (66.7%), TB lymphadenopathy, TB
osteomyelitis and latent TB in 1 (5.6 %) each. Sixteen (47.1%)
patients had contact with an adult suffering from TB of which 7
(43.8%) has drug resistant TB. Three patients (10.3%) out of 29
tested were HIV co-infected. Thirty one (91.2%) children had
received BCG vaccine and 21 out of 25 patients (61.8%) had a
positive mantoux test.
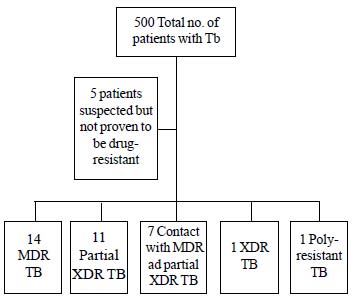 |
|
Fig. 1 Flow diagram of
patients in the study.
|
The individual drug resistance in children to
different drugs was 100% for isoniazid, 97.1% for rifampicin,
76.5% for streptomycin, 67.6% for ethambutol, 47.2% for
ofloxacin, 23.5% for ethionamide, 20.6% for moxifloxacin, and
14.7% for PAS. Repeat TB cultures could only be done in 3
patients of which 2 still grew MTB and one of them showed
additional drug resistance to PAS, kanamycin and ethionamide on
repeat culture.
Factors associated with DR-TB in different
age groups are depicted in Table I. Nine (69.2 %)
boys had contact with an adult suffering from TB as compared to
7 (33.3 %) girls (P=0.045). Type of TB did not differ
between boys and girls (P=0.306). Loss of weight was seen
in 18 (85.7%) girls as compared to 6 (46.2 %) boys (P=0.019).
TABLE I Factors Associated with DR-TB in Different Age Groups
| Factors |
<5 yrs |
5-10 yrs
|
> 10 yrs
|
|
(n=12) |
(n=18)
|
(n=4)
|
|
n (%) |
n (%) |
n (%) |
| Male
|
7 (58.3) |
6 (33.3) |
0 |
| Fever |
7 (58.3) |
15 (83.3) |
2 (50) |
| Cough |
5 (41.7) |
11 (61.1) |
1 (25) |
| Loss of appetite |
7 (58.3) |
13 (72.2) |
3 (75) |
| Loss of weight |
7 (58.3) |
14 (77.8) |
3 (75) |
| Malnourished |
7 (58.3) |
12 (66.7) |
1 (25) |
| Past history of TB |
3 (25) |
11(61.1) |
4 (100) |
| History of TB contact |
8 (66.7) |
7 (38.9) |
1 (25) |
| Type of
drug resistance |
| Contact with Partial
XDR |
2 (16.7) |
1 (5.6) |
0 |
| MDR |
2 (16.7) |
8 (44.4) |
4 (100) |
| MDR with
contact |
3 (25) |
1 (5.6) |
0 |
| XDR |
1 (8.3) |
0 |
0 |
| Partial XDR |
4 (33.3) |
07 (38.9) |
0 |
| Polyresistant |
0 |
1 (5.6) |
0 |
| Type of
current TB |
| Pulmonary TB |
4 (33.3) |
7 (38.9) |
1 (25) |
| Disseminated TB |
2 (16.7) |
7 (38.9) |
1 (25) |
| TB Lymphadenopathy |
2 (16.7) |
1 (5.6) |
1 (25) |
| Abdominal TB |
0 |
2 (11.1) |
1 |
| (25)Latent TB |
1 (8.3) |
1 (5.6) |
0 |
| TB Osteomyelitis |
1 (8.3) |
0 |
0 |
| TBM |
1 (8.3) |
0 |
0 |
| TBM with tuberculoma |
1 (8.3) |
0 |
0 |
Discussion
This is the first study on drug-resistant TB
in children from India. Schaaf, et al. [6] reported a
prevalence of 6.5% of MDR TB in 2003-2005 in children <13 years
of age at Children’s Hospital in South Africa. Recent estimates
on the prevalence of MDR-TB in new smear positive pulmonary TB
(PTB) cases in adults in India is <3% and 12 to 17% amongst
smear positive previously treated PTB cases [1]. In our study,
prevalence of drug resistant TB in children was 6.8%. Though
yield of getting a culture proven drug resistant TB is less in
children as compared to adults due to inability to get the
sputum in children easily, still a prevalence of 6.8% is high
and suggests that prevalence in adults may be even higher.
Schaaf, et al. [6] in their study from
South Africa found that median age of MDR-TB in children was 2.5
years (53.6% boys). In our study, median age of presentation of
DR-TB was 7 years and children in the above 5 years constituted
maximum number of patients. One of the reasons of presentation
of DR-TB in slightly older children as compared to Schaaf, et
al. may be due to inability to get tissue cultures in
younger children [6]. In our study extrapulmonary DR-TB was seen
in 64.7% suggesting that tissue culture diagnosis may be
difficult in these patients. Also most of the patients had been
previously treated for TB or were in contact with an adult
suffering from TB suggesting that the resistance in children may
due to these factors, and thus an older age of presentation.
In our study, the commonest type of drug
resistance was MDR followed by partial XDR. Thus the kind of
DR-TB in our geographical area tends to be multidrug resistant
TB. This is in contrast to study by Alrajhi, et al.
[7] who noted polyresistance in 5% of isolates suggesting
that type of DR-TB tends to vary from area to area. It is
important to identify the trend of resistance pattern in a
community to determine the possible antituberculous therapy that
may be needed in a child. As per WHO statement [3], a patient
can be placed on a likely sensitive regimen based on
epidemiological pattern in that geographical area, while DST
results are pending, to avoid clinical deterioration and prevent
transmission to contacts.
It has been previously reported that HIV
co-infection increases the risk of getting drug resistance TB
[3]. A study conducted in New York city by Gordin, et al.
[8] revealed that HIV and TB co-infected patients were
significantly more likely to develop resistance to at least one
drug (37% vs. 19%) and MDR (19% vs. 6%) than those
without HIV infection. In our study, only 3 patients (10.3%)
were HIV co-infected among 29 tested. However, it is not
possible to comment on whether DR-TB is low in HIV infected
children from this data.
The protective efficacy of BCG for preventing
serious forms of TB [9], is clear but its role to prevent
drug-resistant TB is not known. Routine BCG vaccination is now
recommended for children exposed continually to a patient who
has infectious pulmonary TB caused by M. tuberculosis
strains resistant to isoniazid and rifampin [9]. We cannot
comment on efficacy of BCG on DR-TB from our data.
In a study from Saudi Arabia, Alrajhi, et
al. [7] showed that history of previous ATT was the only
risk factor associated with DR-TB with odds ratio 19.9 (P<
0.001). We also found previous history of tuberculosis and past
history of ATT as an important risk factor for DR-TB in
children, similar to most of the published studies in adults
[9-11]. Previous studies [12,13] have shown that DR-TB infection
rates are more in childhood contacts with resistant index
cases compared with drug-susceptible index cases. However,
the clinical presentation of DR-TB is similar in children of all
ages and does not differ as per the age. This has not been
reported previously and could suggest a similar presentation for
DR-TB across the pediatric age range.
Our study has a limitation as it has small
number of patients, and patients with suspected DR-TB but not
culture proven were not included in the study. Thus, we may have
missed out on some patients who may have had DR-TB. Moreover,
since this is a hospital based study, we may have noted higher
prevalence of DR-TB.
Epidemiological surveillance studies in
children are required to determine the actual prevalence of
drug-resistant TB in children in India. Efforts to isolate TB
bacilli from body fluids or tissues should be aggressively
pursued so that DR-TB can be identified early and patients can
be put on alternative drugs early before significant tissue
damage occurs.
Contributors: Both authors have
contributed, designed and approved the manuscript.
Funding: None; Competing interests:
None stated.
References
1. TB India 2010: RNTCP Status report.
Central TB Division, Directorate General of Health Services,
Ministry of Health and Family Welfare, Nirman Bhawan, New Delhi
- 110001. Available from: http://www.tbcindia.org. Accessed on
24 November, 2010.
2. World Health Organization (WHO).
Anti-Tuberculosis Drug Resistance in the World. Fourth ‘Global
Report. Available at URL:
http://www.who.int/tb/publications/2008/drs_report4_26feb08.pdf.
Accessed on 31December, 2011.
3. World Health Organization (WHO).
Guidelines for the programmatic management of drug-resistant
tuberculosis – 2006. Available at URL:
http://www.tbrieder.org/publications/books_english/who_treatment_mdr.pdf.
Accessed on 31December, 2011.
4. Shah I, Rahangdale A. Partial extensively
drug resistance (XDR) tuberculosis in children. Indian Pediatr.
2011; 48:977-8.
5. World Health Organization (WHO). Guidance
on provider-initiated HIV testing and counselling in health
facilities. Available at URL:
http://www.who.int/hiv/pub/vct/pitc/en/index.html. Accessed on
16February, 2012
6. Schaaf HS. Drug-resistant tuberculosis in
children. S Afr Med J. 2007;97:995-7.
7. Alrajhi AA, Abdulwahab S, Almodovar E, Al-Abdely
HM. Risk factors for drug-resistant Mycobacterium tuberculosis
in Saudi Arabia. Saudi Med J. 2002;23:305-10.
8. Gordin FM, Nelson ET, Matts JP, Cohn DL,
Ernst J, Benator D, et al. The impact of human
immunodeficiency virus infection on drug-resistant tuberculosis.
Am J Respir Crit Care Med. 1996;154:1478-83.
9. The role of BCG vaccine in the prevention
and control of tuberculosis in the United States. A joint
statement by the Advisory Council for the Elimination of
Tuberculosis and the Advisory Committee on Immunization
Practices. MMWR Recomm Rep. 1996;45:1-18.
10. Sharma SK, Turaga KK, Balamurugan A, Saha
PK, Pandey RM, Jain NK, et al. Clinical and genetic risk
factors for the development of multi-drug resistant tuberculosis
in non-HIV infected patients at a tertiary care center in India:
a case-control study. Infect Genet Evol. 2003; 3: 183-188.
11. Espinal MA, Laserson K, Camacho M,
Fusheng Z, Kim SJ, Tlali RE, et al. Determinants of
drug-resistant tuberculosis: analysis of 11 countries. Int J
Tuberc Lung Dis 2001; 5 : 887-893.
12. Snider DE Jr, Kelly GD, Cauthen GM,
Thompson NJ, Kilburn JO. Infection and disease among contacts of
tuberculosis cases with drug-resistant and drug-susceptible
bacilli. Am Rev Respir Dis. 1985;132:125-32.
13. Schaaf HS, Vermeulen HA, Gie RP, Beyers
N, Donald PR. Evaluation of young children in household contact
with adult multidrug resistant pulmonary tuberculosis cases.
Pediatr Infect Dis J. 1999;18:494-500
|

