|
|
|
Indian Pediatr 2011;48:
709-717 |
 |
Revised Statement on Management of Urinary
Tract Infections |
|
Indian Society of Pediatric Nephrology
Correspondence to: Dr M Vijayakumar, Department of
Pediatric Nephrology,
Mehta Children’s Hospitals, Chennai 600 031, India.
Email: [email protected]
|
Justification: In 2001, the Indian Pediatric Nephrology Group
formulated guidelines for management of patients with urinary tract
infection (UTI). In view of emerging scientific literature, the
recommendations have been reviewed.
Process: Following a preliminary meeting in
November 2010, a document was circulated among the participants to arrive
at a consensus on the evaluation and management of these patients.
Objectives: To revise and formulate guidelines on
management of UTI in children.
Recommendations: The need for accurate diagnosis of
UTI is emphasized due to important implications concerning evaluation and
follow up. Details regarding clinical features and diagnosis, choices and
duration of therapy and protocol for follow up are discussed. UTI is
diagnosed on a positive culture in a symptomatic child, and not merely by
the presence of leukocyturia. The need for parenteral therapy in UTI in
young infants and those showing toxicity is emphasized. Patients with
aysmptomatic bacteriuria do not require treatment. The importance of bowel
bladder dysfunction in the causation of recurrent UTI is highlighted.
Infants with the first UTI should be evaluated with micturating
cystourethrography. Vesicoureteric reflux (VUR) is initially managed with
antibiotic prophylaxis. The prophylaxis is continued till 1 year of age in
patients with VUR grades I and II, and till 5 years in those with higher
grades of reflux or until it resolves. Patients and their families are
counselled about the need for early recognition and therapy of UTI.
Children with VUR should be followed up with serial ultrasonography and
direct radionuclide cystograms every 2 years, while awaiting resolution.
Siblings of patients with VUR should be screened by ultrasonography.
Children with renal scars need long term follow up on yearly basis for
growth, hypertension, proteinuria, and renal size and function.
Key words: Child, India, Prevention, Urinary tract infections,
Vesicoureteric reflux.
|
|
Urinary tract infection (UTI) is a common bacterial infection in infants
and children. The risk of having a UTI before the age of 14 years is
approximately 1-3% in boys and 3-10% in girls [1,2]. The diagnosis of UTI
is often missed in infants and young children, as urinary symptoms are
minimal and often non-specific. Rapid evaluation and treatment of UTI is
important to prevent renal parenchymal damage and renal scarring that can
cause hypertension and progressive renal damage [3]. Pediatricians should
be aware of the clinical features, diagnosis, management and evaluation of
children with UTI. Even a single confirmed UTI should be taken seriously,
especially in young children, due to the potential for renal parenchymal
damage.
An Expert Group Meeting of the Indian Society of
Pediatric Nephrology was held on 12 th
November, 2010 in Kolkata to review the guidelines published in Indian
Pediatrics in 2001 [4]. New evidence was analyzed, with an aim to
update and revise the guidelines. The revisions are highlighted in
Table I.
TABLE I Major Revisions in This Document
-
The importance of urine culture on a
correctly collected specimen is reemphasized. The diagnosis of
urinary tract infection (UTI) must be based on a positive urine
culture.
-
Patients with UTI should be evaluated for the
presence of complications, underlying anomalies or voiding
dysfunction.
-
Recommendations on imaging following the
first episode of UTI are revised. Detailed investigations are
done in infants. In older children, micturating
cystourethrography is done in those who show abnormalities on
ultrasonography and DMSA scintigraphy.
-
Patients with recurrent UTI and/or
vesicoureteric reflux should be evaluated for bowel bladder
dysfunction.
-
Patients with grades I and II reflux should
receive antibiotic prophylaxis till they are 1 year old. Those
with higher grades of reflux are given prophylaxis till 5 years
of age, or longer in case of bowel bladder dysfunction or
breakthrough UTI.
|
Definitions
Infection of the urinary tract is identified by growth
of a significant number of organisms of a single species in the urine, in
the presence of symptoms. The diagnosis of UTI should be made only in
patients with a positive urine culture, since this has implications for
detailed evaluation and follow up. Recurrent UTI, defined as the
recurrence of symptoms with significant bacteriuria in patients who have
recovered clinically following treatment, is common in girls. Recurrent
UTI add to parental anxiety, medical costs and the risk of renal
parenchymal damage in young infants.
Clinical Features
UTI is an important cause for fever without a focus,
especially in children less than 2 years old [5,6]. In neonates, UTI is
usually a part of septicemia and presents with fever, vomiting, lethargy,
jaundice and seizures. Infants and young children present with recurrent
fever, diarrhea, vomiting, abdominal pain and poor weight gain. Older
children show fever, dysuria, urgency, frequency and abdominal or flank
pain. Adolescents may have symptoms restricted to the lower tract, and
fever may not be present.
The distinction between upper and lower UTI is
difficult and not necessary. In view of risks of renal parenchymal damage
associated with delayed treat-ment, UTI in children is considered to
involve the upper tract and should be treated promptly. Patients with
features of systemic toxicity are considered as having complicated UTI,
while those without these features are referred to as simple UTI (Table
II) [4]. This distinction has implications for therapy, as is
discussed later.
| Significant bacteriuria |
Colony count of >105/mL of a single
species in a midstream clean catch sample. |
| Asymptomatic bacteriuria |
Significant bacteriuria in the absence of symptoms of
urinary tract infection (UTI). |
| Simple UTI |
UTI with low grade fever, dysuria, frequency, and
urgency; and absence of symptoms of complicated UTI. |
| Complicated UTI |
Presence of fever >39ºC, systemic toxicity, persistent
vomiting, dehydration, renal angle tenderness and raised creatinine. |
| Recurrent infection |
Second episode of UTI. |
Diagnosis
The diagnosis of UTI is based on positive culture of a
properly collected specimen of urine. While urinalysis enables a
provisional diagnosis of UTI, a specimen must be obtained for culture
prior to therapy with antibiotics [7].
Significant pyuria is defined as >10 leukocytes per mm 3
in a fresh uncentrifuged sample, or >5 leukocytes per high power field in
a centrifuged sample. Leukocyturia might occur in conditions such as
fever, glomerulonephritis, renal stones or presence of foreign body in the
urinary tract. The detection of leukocyturia in absence of significant
bacteriuria is not sufficient to diagnose a UTI. Rapid dipstick based
tests, which detect leukocyte esterase and nitrite, are useful in
screening for UTI. A combination of these tests has moderate sensitivity
and specificity for detecting UTI, and is diagnostically as useful as
microscopy [1].
Collection of specimen for culture
A clean-catch midstream specimen is used to minimize
contamination by periurethral flora. Contamination can be minimized by
washing the genitalia with soap and water. Antiseptic washes and forced
retraction of the prepuce are not advised. In neonates and infants, urine
sample is obtained by either suprapubic aspiration or transurethral
bladder catheterization. Both techniques are safe and easy to perform [7].
The urine specimen should be promptly plated within one
hour of collection. If delay is anticipated, the sample can be stored in a
refrigerator at 4ºC for up to 12-24 hours. Cultures of specimens collected
from urine bags have high false positive rates, and are not recommended.
A urine culture should be repeated in case
contamination is suspected, e.g., mixed growth of two or more
pathogens, or growth of organisms that normally constitute the
periurethral flora (lacto-bacilli in healthy girls; enterococci in infants
and toddlers). The culture should also be repeated in situations where UTI
is strongly suspected but colony counts are equivocal. The number of
bacteria required for defining UTI depends on the method of urine
collection (Table III) [2,4,5].
TABLE III Criteria For The Diagnosis of UTI
|
Method of collection |
Colony count |
Probability of infection |
| Suprapubic aspiration |
Any number of pathogens |
99% |
| Urethral
catheterization |
>5 × 104 CFU/mL |
95% |
| Midstream clean catch |
>105 CFU/mL |
90-95% |
|
CFU: colony forming units. |
Initial Evaluation
The patient is examined for the degree of toxicity,
dehydration and ability to retain oral intake. The blood pressure should
be recorded and history regarding bowel and bladder habits elicited. The
child is examined for features that suggest an underlying functional or
urological abnormality (Tables IV and V). Complete
blood counts, serum creatinine and a blood culture should be done in
infants and children with complicated UTI.
TABLE IV Features Suggesting Underlying Structural Abnormality
| Distended bladder |
| Palpable, enlarged kidneys |
| Tight phimosis; vulval synechiae |
| Palpable fecal mass in the colon |
| Patulous anus; neurological deficit in lower limbs |
| Urinary incontinence |
| Previous surgery of the urinary tract, anorectal
malformation or meningomyelocele |
Table V Features Suggestive of Bowel Bladder Dysfunction
| Recurrent urinary tract infections |
| Persistent high grade vesicoureteric reflux |
| Constipation, impacted stools |
| Maneuvers to postpone voiding (holding maneuvers,
e.g., Vincent curtsy, squatting) |
| Voiding less than 3 or more than 8 times a day |
| Straining or poor urinary stream |
| Thickened bladder wall >2 mm |
| Post void residue >20 mL |
| Spinning top configuration of bladder on micturating
cystourethrogram |
Immediate Treatment
The patient’s age, features suggesting toxicity and
dehydration, ability to retain oral intake and the likelihood of
compliance with medication(s) help in deciding the need for
hospitalization. Therapy should be prompt to reduce the morbidity of
infection, minimize renal damage and subsequent complications.
Children less than 3 months of age and those with
complicated UTI should be hospitalized and treated with parenteral
antibiotics. The choice of antibiotic should be guided by local
sensitivity patterns. A third generation cephalosporin is preferred (Table
VI). Therapy with a single daily dose of an aminoglycoside may be
used in children with normal renal function [8]. Once the result of
antimicrobial sensitivity is available, the treatment may be modified.
Intravenous therapy is given for the first 2-3 days followed by oral
antibiotics once the clinical condition improves.
TABLE VI Antimicrobials for Treatment of UTI
| Medication |
Dose, mg/kg/day |
| Parenteral |
| Ceftriaxone |
75-100, in 1-2 divided doses IV |
| Cefotaxime |
100-150, in 2-3 divided doses IV |
| Amikacin |
10-15, single dose IV or IM |
| Gentamicin |
5-6, single dose IV or IM |
| Coamoxiclav |
30-35 of amoxicillin, in 2 divided doses IV |
| Oral |
| Cefixime |
8-10, in 2 divided doses |
| Coamoxiclav |
30-35 of amoxicillin, in 2 divided doses |
| Ciprofloxacin |
10-20, in 2 divided doses |
| Ofloxacin |
15-20, in 2 divided doses |
| Cephalexin |
50-70, in 2-3 divided doses |
Children with simple UTI and those above 3 months of
age are treated with oral antibiotics (Table VI). With
adequate therapy, there is resolution of fever and reduction of symptoms
by 48-72 hours. Failure to respond may be due to presence of resistant
pathogens, complicating factors or noncompliance; these patients require
re-evaluation.
Duration of Treatment
The duration of therapy is 10-14 days for infants and
children with complicated UTI, and 7-10 days for uncomplicated UTI [4, 5].
Adolescents with cystitis may be treated with shorter duration of
antibiotics, lasting 3 days [1]. Following the treatment of the UTI,
prophylactic antibiotic therapy is initiated in children below 1 year of
age, until appropriate imaging of the urinary tract is completed.
Supportive Therapy
During an episode of UTI, it is important to maintain
adequate hydration. A sick, febrile child with inadequate oral intake or
dehydration may require parenteral fluids. Routine alkalization of the
urine is not necessary. Paracetamol is used to relieve fever; therapy with
non steroidal anti- inflammatory agents should be avoided. A repeat urine
culture is not necessary, unless there is persistence of fever and
toxicity despite 72 hours of adequate antibiotic therapy.
Evaluation after the first UTI
The aim of investigations is to identify patients at
high risk of renal damage, chiefly those below one year of age, and those
with VUR or urinary tract obstruction. Evaluation includes ultrasonography,
dimercaptosuccinic acid (DMSA) renal scan and micturating
cystourethrography (MCU) performed judiciously as shown in Fig.
1. An ultrasonogram provides information on kidney size, number and
location, presence of hydronephrosis, urinary bladder anomalies and
post-void residual urine. DMSA scintigraphy is a sensitive technique for
detecting renal parenchymal infection and cortical scarring. MCU detects
VUR and provides anatomical details regarding the bladder and the urethra.
Follow-up studies in patients with VUR can be performed using direct
radionuclide cystography.
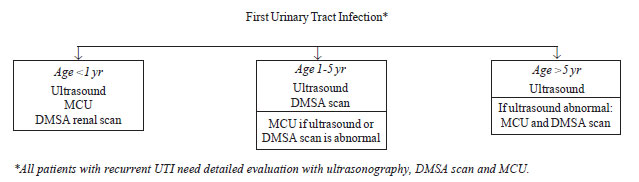 |
|
Fig. 1 Evaluation following initial urinary
tract infection. MCU: micturating cystourethrogram; DMSA
dimercaptosuccinic acid. |
There is limited evidence that intensive imaging and
subsequent management alters the long-term outcome of children with reflux
nephropathy diagnosed following a UTI. With availability of antenatal
screening, most important anomalies have already been detected and managed
after birth. Therefore, there is considerable debate regarding the need
and intensity of radiologic evaluation in children with UTI [1,9].
The Expert Group reviewed the current literature,
keeping in view that in our country the diagnosis of UTI is often missed
or delayed, and there are limitations of infrastructure and scarcity of
resources for routine antenatal screening. Based on the above, it
concluded that all children with the first UTI should undergo radiological
evaluation. The detection of significant scarring, high grade VUR or
obstructive uropathy might enable interventions that prevent progressive
kidney damage in the long-term. Since infants and young children are at
the highest risk for renal scarring, it is necessary that this group
undergo focused evaluation.
It is recommended that all infants with UTI be screened
by ultrasonography, followed by MCU and DMSA scintigraphy. Since older
patients (1-5 year old) with significant reflux and scars or urinary tract
anomalies are likely to show abnormalities on ultrasonography or
scintigraphy, a MCU is advised in patients having abnormalities on either
of the above investigations. Children older than 5 years are screened by
ultrasonography and further evaluated only if this is abnormal.
It is emphasized that patients with recurrent UTI at
any age should undergo detailed imaging with ultrasonography, MCU and DMSA
scintigraphy.
Ultrasonography should be done soon after the diagnosis
of UTI. The MCU is recommended 2-3 weeks later, while the DMSA scan is
carried out 2-3 months after treatment. An early DMSA scan, performed soon
after a UTI, is not recommended in routine practice. Patients showing
hydronephrosis in the absence of VUR should be evaluated by diuretic
renography using 99mTc-labeled
diethylenetriamine-pentaacetic acid (DTPA) or mercaptoacetylglycine
(MAG-3). These techniques provide quantitative assessment of renal
function and drainage of the dilated collecting system.
Prevention of Recurrent UTI
General
Adequate fluid intake and frequent voiding is advised;
constipation should be avoided [2,5]. In children with VUR who are toilet
trained, regular and volitional low pressure voiding with complete bladder
emptying is encouraged. Double voiding ensures emptying of the bladder of
post void residual urine. Circumcision reduces the risk of recurrent UTI
in infant boys, and might therefore have benefits in patients with high
grade reflux [10,11].
Bowel bladder dysfunction
Children presenting with recurrent UTI or persistent
VUR often have an associated voiding disorder, which are characterized by
abnormal patterns of micturition in presence of intact neuronal pathways
without congenital or anatomical abnormalities. Abnormal bladder pressure
and urinary stasis predispose these children to recurrent UTI. There may
be an abnormality either during the filling phase as in an overactive
bladder, or the evacuation phase as in dysfunctional voiding [12]. Since
constipation is often associated with a functional voiding disorder,
the condition is referred to as Bowel bladder dysfunction (BBD). Children
with recurrent UTI are likely to have dysfunctional voiding [5,13].
Features suggestive of voiding disorders are shown in Table V.
Evaluation for a voiding disorder includes a record of
frequency and voided volume and fluid intake for two to three days. It is
useful to watch the urinary stream, and for post void dribbling in boys.
Urodynamic studies are done in selected cases. The management of voiding
disorders should be carried out in collaboration with an expert. This
includes the exclusion of neurological causes, institution of structured
voiding patterns and management of constipation. In patients with an
overactive bladder, therapy with anticholinergic medications (e.g.,
oxybutinin) is effective. Patients with bowel bladder dysfunction and
large post void residues, benefit from timely voiding, bladder retraining,
and clean intermittent catheterization.
Antibiotic Prophylaxis
Long-term, low dose, antibacterial prophylaxis is used
to prevent recurrent, febrile UTI (Table VII). The
antibiotic used should be effective, non-toxic with few side effects and
should not alter the growth of commensals or induce bacterial resistance
[14].
TABLE VII Antimicrobials for Prophylaxis of Urinary Tract Infections
|
Medication |
Dose, mg/kg/day |
Remarks |
|
Cotrimoxazole |
1-2* |
Avoid in infants <3 mo, glucose-6-phosphate dehydrogenase deficiency |
|
Nitrofurantoin |
1-2 |
May cause vomiting and nausea; avoid in infants <3 mo, G6PD
deficiency, renal insufficiency |
|
Cephalexin |
10 |
Drug of choice in first 3-6 mo of life |
|
Cefadroxil |
5 |
An alternative agent in early infancy |
|
Usually given as
single bedtime dose; *of trimethoprin. |
Indications and Duration of Prophylaxis
The indications and duration of prophylaxis depend on
patient age and presence or absence of VUR. Antibiotic prophylaxis is
recommended for patients with (i) UTI below 1-yr of age,
while awaiting imaging studies, (ii) VUR (see Table VIII),
(iii) frequent febrile UTI (3 or more episodes in a year) even if
the urinary tract is normal [14, 15]. Antibiotic prophylaxis is not
advised in patients with urinary tract obstruction (e.g., posterior
urethral valves), urolithiasis and neurogenic bladder, and in patients on
clean intermittent catheterization.
TABLE VIII Management of Vesicoureteric Reflux
| VUR grade |
Management |
| Grades I and II |
Antibiotic prophylaxis until 1 yr old. Restart
antibiotic prophylaxis if breakthrough febrile UTI. |
| Grades III to V |
Antibiotic prophylaxis up to 5 yr of age. Consider
surgery if breakthrough febrile UTI. |
| |
Beyond 5 yr: Prophylaxis continued if there is bowel
bladder dysfunction. |
Breakthrough UTI on Prophylactic Antibiotics
Breakthrough UTI results either from poor compliance or
associated voiding dysfunction. The UTI should be treated with appropriate
antibiotics. A change of the medication being used for prophylaxis is
usually not necessary. There is no role for cyclic therapy, where the
antibiotic used for prophylaxis is changed every 6-8 weeks.
Asymptomatic Bacteriuria
Asymptomatic bacteriuria is the presence of significant
bacteriuria in the absence of symptoms of UTI. Its frequency is 1-2% in
girls and 0.2% in boys [1]. Asymptomatic bacteriuria is a benign
condition, which does not cause renal injury and requires no treatment.
The organism isolated in most instances is E. coli, which is of low
virulence. Eradication of these organisms is often followed by symptomatic
infection with more virulent strains. Therapy of asymptomatic bacteriuria
or antibiotic prophylaxis is not required [1].
The presence of asymptomatic bacteriuria in a patient
previously treated for UTI should not be considered as recurrent UTI.
Vesicoureteric Reflux
VUR is seen in 40-50% infants and 30-50% children with
UTI, and resolves with age. Its severity is graded using the International
Study Classification from grade I to V, based on the appearance of the
urinary tract on MCU [16]. Lower grades of reflux (grade I-III) are more
likely to resolve. Secondary VUR is often related to bladder outflow
obstruction, as with posterior urethral valves, neurogenic bladder or a
functional voiding disorder.
The presence of moderate to severe VUR, particularly if
bilateral, is an important risk factor for pyelonephritis and renal
scarring, with subsequent risk of hypertension, albuminuria and
progressive kidney disease. The risk of scarring is highest in the first
year of life [17]. The presence of intrauterine VUR has been associated
with renal hypoplasia or dysplasia [18].
Therapy for Primary VUR
Over the last decade it has been increasingly
recognized that not all children with VUR benefit from diagnosis or
treatment. In some patients the reflux is innocuous and self limiting. In
others, VUR is accompanied with renal damage that has an onset during the
intrauterine period with dysplastic kidneys at birth, where the treatment
of VUR will not change the long term outcome [18]. There is a subset of
children who would benefit from treatment; however, identifying this group
of patients remains a challenge.
Conventional therapy for VUR includes antibiotic
prophylaxis and surgical intervention [11, 19, 20]. A recent systematic
review on patients with dilating reflux concluded that the outcomes
following surgical repair versus prophylaxis were similar in terms
of the number of breakthrough UTI and risk of renal scarring [20].
Experts recommend that the management of patients with VUR should depend
on the patient age, grade of reflux and whether there are any breakthrough
infections [11].
The proposed guidelines for management of VUR are
outlined in Table VIII. It is recommended that patients
should initially receive antibiotic prophylaxis while awaiting spontaneous
resolution of VUR. A close follow up is required for occurrence of
breakthrough UTI. Repeat imaging is required after 18-36 months in
patients with grade III-V VUR. Radionuclide cystogram, with lower
radiation exposure, has higher sensitivity for detecting reflux and is
therefore preferred for follow-up evaluation. Since the risk of recurrent
UTI and renal scarring is low after 4-5 years of age [11, 21], it is
advised that prophylaxis be discontinued in children older than 5 years
with normal bowel and voiding habits, even if mild to moderate reflux
persists.
While evidence from few studies suggests that the
strategy of prompt diagnosis and treatment of UTI might be as effective as
antibiotic prophylaxis [1,22], this approach requires validation in
controlled trials. The clinician should discuss the benefits and risks of
withholding antibiotic prophylaxis with the parents [19, 20].
Patients with grade III to V reflux may be offered
surgical repair if they have breakthrough febrile UTI, if parents prefer
surgical intervention to prophylaxis, or in patients who show
deterioration of renal function [20,21]. An evaluation for voiding
dysfunction (based on history, voiding diary) should be done before
surgery. Antibiotic prophylaxis is continued for 6 months after surgical
repair.
The availability of dextranomer/hyaluronic acid
copolymer (Deflux) endoscopic treatment has been proposed as an
alternative to surgical repair for patients with VUR [23]. While results
are satisfactory in surgeons experienced with the procedure, a significant
proportion of patients, particularly those with bowel bladder dysfunction,
may show persistence and/or recurrence of reflux and progressive renal
damage [24,25]. In view of limited prospective randomized controlled
trials, the use of endoscopic correction is currently not recommended as
first line therapy [11].
Screening of siblings and offspring
Reflux is inherited in an autosomal dominant manner
with incomplete penetrance; 27% siblings and 35% offspring of patients
show VUR [26]. Ultrasonography is recommended to screen for the presence
of reflux. Further imaging is required if ultrasonography is abnormal [11,
26].
Long Term Follow-up
Patients with a renal scar (reflux nephropathy) are
counseled regarding the need for early diagnosis and therapy of UTI and
regular follow up. Physical growth and blood pressure should be monitored
every 6-12 months, through adolescence. Investi-gations include urinalysis
for proteinuria and estimation of blood levels of creatinine. Annual
ultrasound examinations are done to monitor renal growth.
TABLE IX Indication for Referral to a Pediatric Nephrologist
|
• |
Recurrent urinary tract infections |
|
• |
Urinary tract infections in association with bowel bladder
dysfunction |
|
• |
Patients with vesicoureteric reflux |
|
• |
Underlying urologic or renal abnormalities |
|
• |
Children with renal scar, deranged renal functions, hypertension |
Indications for a referral to a Pediatric Nephrologist
UTI can be effectively managed by the primary care
physician. However because of their potential for renal parenchymal
damage, scarring and subsequent chronic kidney disease, patients having
risk factors that increase the likelihood of complications should be
managed in collaboration with an expert (Table IX).
Writing Committee: M Vijayakumar, M Kanitkar, BR
Nammalwar and Arvind Bagga.
Acknowledgment: The Committee acknowledges the
contributions of A Sinha, S Uthup, P Hari, A Iyengar, A Pahari, M Shah and
S Banerjee.
Participants of the Expert Group Meeting held on 12
November 2010 at Kolkata: Aditi Sinha, Anand S Vasudev, Arpana
Iyengar, Arvind Bagga, Ashima Gulati, BR Nammalwar, H Lekha, Indira
Agarwal, Jayati Sengupta, Jyoti Sharma, Kamini Mehta, Kishore Phadke,
Kumud P Mehta, Madhuri Kanitkar, Manoj G Matnani, Madhusmita Sengupta,
Mehul Shah, Pankaj Hari, Prabha Senguttuvan, Prahlad N, Premalatha, Preeti
Shanbag, RN Srivastava, Sanjeev Gulati, Saroj K Patnaik, Sidharth K Sethi,
Susan Uthup, Tamilarasi V, Tathagata Bose, Uma S. Ali, Vijayakumar M (convener),
Vinay K Agarwal and VK Sairam.
References
1. National Collaborating Centre for Women’s and
Children’s Health. Urinary tract infection in children diagnosis,
treatment and long-term management. RCOG Press, London 2007. Available
from www.rcpch.ac.uk/Research/ce/Clinical-Audit/Urinary-Tract-Infection,
Accessed on 17 March, 2011.
2. Chang SL, Shortliffe LD. Pediatric urinary tract
infections. Pediatr Clin North Am. 2006;53:379-400.
3. Smellie JM, Prescod NP, Shaw PJ, Risdon RA, Bryant
TN. Childhood reflux and urinary infection: a follow-up of 10–41 years in
226 adults. Pediatr Nephrol. 1998;12:727-36.
4. Indian Pediatric Nephrology Group. Consensus
statement on management of urinary tract infections. Indian Pediatr.
2001;38:1106-15.
5. American Academy of Pediatrics, Committee on Quality
Improvement, Subcommittee on Urinary Tract Infections. Practice
parameters: The diagnosis, treatment and evaluation of the initial urinary
tract infections in febrile infants and young children. Pediatrics.
1999;103:843-52.
6. Nammalwar BR, Vijayakumar M, Sankar J, Ramnath B,
Prahlad N. Evaluation of the use of DMSA in culture positive UTI and
culture negative acute pyelonephritis. Indian Pediatr. 2005;42:691-6.
7. Srivastava RN, Bagga A. Urinary tract infection.
In: Pediatric Nephrology, 5th edn. New Delhi: Jaypee Brothers; 2011.p.
273-300.
8. Bloomfield P, Hodson EM, Craig JC. Antibiotics for
acute pyelonephritis in children. Cochrane Database Syst Rev.
2003;3:CD003772.
9. Lim R. Vesicoureteral reflux and urinary tract
infection: evolving practices and current controversies in pediatric
imaging. Am J Roentgenol. 2009;192:1197-1208.
10. Singh-Grewal D, Macdessi J, Craig J. Circumcision
for the prevention of urinary tract infection in boys: a systematic review
of randomized trials and observational studies. Arch Dis Child.
2005;90:853-8.
11. Peters CA, Skoog SJ, Arant BS, Copp HL, Elder JS,
Hudson RG, et al; American Urological Association Education and
Research, Inc. Summary of the AUA guideline on management of primary
vesicoureteral reflux in children. J Urol. 2010;184:1134-44.
12. Nevéus T, von Gontard A, Hoebeke P, Hjälmås K,
Bauer S, Bower W, et al. The standardization of terminology of
lower urinary tract function in children and adolescents: report from the
standardization committee of the International Children’s Continence
Society. J Urol. 2006;176:314-24.
13. Ramamurthy HR, Kanitkar M. Recurrent urinary tract
infection and functional voiding disorders. Indian Pediatr.
2008;45:689-91.
14. Mangiarotti P, Pizzini C, Fanos V. Antibiotic
prophylaxis in children with relapsing urinary tract infections: review. J
Chemother. 2000;12:115-23.
15. Dai B, Liu Y, Jia J, Mei C. Long-term antibiotics
for the prevention of recurrent urinary tract infection in children: a
systematic review and meta-analysis. Arch Dis Child. 2010;95:499-508.
16. Lebowitz RL, Olbing H, Parkkulainen KV, Smellie JM,
Tamminen-Mobius TE. International system of radiographic grading of
vesicoureteric reflux: International Reflux Study in Children. Pediatr
Radiol. 1985;15:105-9.
17. Ylinen E, Ala-Houhala M, Wikstrom S. Risk of renal
scarring in vesicoureteral reflux detected either antenatally or during
the neonatal period. Urology. 2003;61:1238-42.
18. Craig JC, Irwig LM, Knight JF, Roy LP. Does
treatment of vesicoureteric reflux in childhood prevent end-stage renal
disease attributable to reflux nephropathy? Pediatrics. 2000;105:1236-41.
19. Williams GJ, Lee A, Craig JC. Long-term antibiotics
for preventing recurrent urinary tract infection in children. Cochrane
Database Syst Rev. 2006;3:CD001534.
20. Hodson EM, Wheeler DM, Smith GH, Craig JC,
Vimalachandra D. Interventions for primary vesicoureteric reflux. Cochrane
Database Syst Rev. 2007;3:CD 001532.
21. Greenbaum LA, Mesrobian HGO. Vesicoureteral reflux.
Pediatr Clin North Am. 2006;53:413-27.
22. Garin EH, Olavarria F, Garcia NV, Valenciano B,
Campos A, Young L. Clinical significance of primary vesicoureteral reflux
and urinary antibiotic prophylaxis after acute pyelonephritis: a
multicenter, randomized, controlled study. Pediatrics. 2006;117:626-32.
23. Molitierno JA, Scherz HC, Kirsch AJ. Endoscopic
treatment of vesicoureteral reflux using dextranomer hyaluronic acid
copolymer. J Pediatr Urol. 2008;4:221-8.
24. Lee EK, Gatti JM, Demarco RT, Murphy JP. Long-term
follow up of dextranomer/ hyaluronic acid injection for vesicoureteral
reflux: late failure warrants continued follow up. J Urol.
2009;181:1869-74.
25. Holmdahl G, Brandström P, Läckgren G, Sillén U, Stokland
E, Jodal U, et al. The Swedish reflux trial in children: II.
Vesicoureteral reflux outcome. J Urol. 2010; 184: 280-5.
26. Skoog SJ, Peters CA, Arant BS, Copp HL, Elder JS,
Hudson RG, et al; American Urological Association Education and
Research. Pediatric vesicoureteral reflux guidelines panel summary report:
clinical practice guidelines for screening siblings of children with
vesicoureteral reflux and neonates/infants with prenatal hydronephrosis. J
Urol. 2010;184:1145-51.
|
|
|
 |
|

