|
|
|
Indian Pediatr 2013;50:
1025-1032 |
 |
Variability of Thinness and its Relation to
Cardio-metabolic Risk Factors using Four Body Mass Index
References in School-children from Delhi, India
|
|
1,8 Pankaj Garg,
2,9Supreet Kaur,
3,10Dileep Gupta,
4Clive Osmond,
5Ramakrishnan Lakshmy,
11Shikha Sinha,
6 Umesh Kapil,
7 HPS Sachdev*
From the 1, 3, 7,11Department of Pediatrics and
Clinical Epidemiology, Sitaram Bhrtia Institute of Science and Research,
B-16, Qutab Institutional Area, New Delhi 110016, India; 2, 6Departments
of Human Nutrition and 5 Cardiac Biochemistry, All India
Institute of Medical Sciences, New Delhi 110029, India; 4Medical
Research Council Lifecourse Epidemiology Unit, University of
Southampton, Southampton General Hospital, Southampton S016 6YD, UK;
8Current affiliation: Conjoint Associate Lecturer, School of
Women’s and Children Health, University of New South Wales and
Department of Community Pediatrics, Liverpool Hospital, NSW, Australia;
9Current affiliation: Senior Programme Associate, Public
Health Foundation of India (PHFI), Vasant Kunj, New Delhi 110070 and
10Current affiliation: Statistical Officer, Planning
Department, Government of NCT of Delhi, New Delhi 110002.
Correspondence to: Prof HPS Sachdev, Senior Consultant
Pediatrics and Clinical Epidemiology, E- 6/12, Vasant Vihar, New Delhi
110 057, India.
Email: [email protected]
Published online: June 5, 2013; PII: S097475591200871
|
Objectives: To compare: (i) prevalences of thinness in
school-children by four body mass index references in common use viz.,
Centre for Disease Control (CDC); Cole; Indian Academy of Pediatrics
(IAP); World Health Organization (WHO); and (ii) relationship of
thinness with absence of cardio-metabolic risk factors in these BMI
references.
Design: Cross-sectional.
Setting: Schools in Delhi.
Participants: Anthropometry and blood pressure
were measured in 16,245 school children aged 5 to 18 years. Fasting
lipids and blood sugar were estimated in 2796 subjects.
Outcome measures: Age and sex-specific
prevalences of thinness and predictive ability of reference cut-off for
detecting any cardio-metabolic risk factor were compared.
Results: Prevalence of thinness varied with the
reference employed; more so for boys. Overall prevalence of thinness was
least with IAP reference and highest with CDC cut-offs (6.6% to 16.9% in
boys, 6.5% to 10.3% in girls). Children identified as thin by any
reference had comparable, significantly lower risks (OR 0.59 to 0.73) of
associated cardio-metabolic aberrations. In subjects with any
cardio-metabolic or blood pressure aberration, the prevalence of
thinness was highest with CDC and least with IAP definition.
Conclusion: Prevalence of thinness varies
considerably with the reference employed. Thin children, identified by
any reference, have a lower risk of associated cardio-metabolic
aberrations; however, thinness is a poor diagnostic test for this
purpose. In populations undergoing nutrition transition, there is a need
to link cardio-metabolic risk factors with recommended anthropometric
criteria to define undernutrition.
Keywords: Body mass index, Cholesterol, Glucose, Hypertension,
Thinness, Triglyceride.
|
|
Adults below a body mass index
(BMI) of 18.5 kg/m
2 are
classified as thin or underweight [1,2]. This concept has been
extrapolated to adolescents and children to develop gender and
age-specific BMI cut-offs to assess prevalence of thinness [2,3] in
various settings, Centre for Disease Control (CDC) and World Health
Organization (WHO) BMI charts are also available for this purpose [4,5].
The availability of multiple cut-offs based on different BMI references
poses a challenge both for clinical practice and for quantification of
public health burden. Considerable variations in prevalences of obesity
(9.3% to 21% in boys, 4.1% to 35% in girls) and metabolic syndrome
abnormalities in anthropometrically obese children are documented with
use of different BMI references in various settings [6-11].
Considerable variations may likewise occur for
thinness, even in low and middle income countries (LMICs).
The National Family Health Survey (NFHS-3) data from
India has documented a high prevalence of undernutrition in children
younger than five years using WHO multi-country growth reference
[12,13]. However, there is paucity of similar nationally representative
data in older children and adolescents. The estimated burden of thinness
(undernutrition) from future, relevant data sets may be substantially
influenced by the choice of growth standard. It is therefore important
to understand and quantify the variations in prevalences of thinness, if
any with the commonly employed, international and national, BMI
references in older children and adolescents.
In LMICs, anthropometrically undernourished children
are often prescribed food supplementation to improve body size. However,
cardio-metabolic risk factors (proxy for overnutrition) can also
theoretically occur in a proportion of anthropometrically undernourished
children, especially in settings of rapid nutrition transition. In the
short-term, food supplementation may worsen cardio-metabolic risk
factors in such individuals. In the long term, if this intervention
leads to adult overweight or obesity, the propensity to develop chronic
diseases and hypertension will increase [14-16]. It is therefore also
important to determine if thinness predicts absence of cardio-metabolic
risk factors and whether this relationship alters with different BMI
references.
This study was therefore designed to compare: (i)
prevalences of thinness in school-children by four BMI references in
common use, namely, Cole, et al. [2], WHO [4], CDC [5] and Indian
Academy of Pediatrics (IAP)
[17]; and (ii) relationship of thinness with absence of
cardio-metabolic risk factors in these BMI references.
Methods
This cross-sectional study was conducted between
January 2005 and March 2007. All the public and private schools
in National Capital Territory of Delhi were enlisted and stratified into
low (LIG), middle (MIG) and high (HIG) income groups on the basis of
fees charged. Government and Municipal Corporation of Delhi schools were
considered as LIG, Kendriya Vidhalayas as MIG and private schools
charging monthly tuition fees above Rs.1000 as HIG. Probability
proportionate to size (PPS) sampling methodology was utilized for
selection of 90 schools. In each of the socio-economic group, 30
clusters were selected. We evaluated 180 ± 10 subjects, between 5 and 18
years of age, from each cluster. Written informed consent was taken from
the school authorities and parents. Institutional ethics approval was
granted by the All India Institute of Medical Sciences, New Delhi.
Weight and height were recorded using standard
methodology [18]. Weight was recorded on SECA electronic weighing scale
to the nearest 100g. Height was recorded on anthropometric height board
to the nearest 0.1cm. Three readings of height and weight were taken,
and the mean of the last two readings was recorded. Systolic and
diastolic blood pressures after a period of 10 minutes rest were
measured in sitting position with an appropriate sized cuff on a mercury
sphygmomanometer. Three readings were taken, and their mean was
considered as the individual’s blood pressure. All these measurements
were recorded by trained nutritionists.
In consenting subjects, 5mL of fasting blood sample
was transported to laboratory for estimating serum total cholesterol,
triglyceride, high density lipoprotein (HDL) and sugar. For quality
assurance, three levels of internal controls were run with each batch of
twenty samples. The intra-assay and inter-assay variation was
less than 2% and 3%, for cholesterol and triglyceride levels,
respectively; and less than 2.5% and 3.5% for HDL cholesterol and sugar,
respectively. Estimation of total serum cholesterol was done by CHOD-PAP
(cholesterol oxidase/p-aminophenazone) method and triglycerides by
GPO-PAP (glycerolphosphate oxidase–peroxidase amino-phenazone) method
[19,20]. High-density lipoprotein cholesterol (HDL) was estimated by the
precipitation method using phosphotungstate/magnesium precipitation of
apolipoprotein B containing lipoproteins followed by the estimation of
cholesterol in the supernatant by enzymatic method. Low density
lipoprotein (LDL) was calculated using Friedewald formula [21].
Fasting glucose was estimated using the standard
glucose oxidase method. Estimations of all these biochemical parameters
were available in 2796 children (LIG – 626, MIG – 962, HIG – 1208).
Pre-hypertension was defined as systolic and
diastolic blood pressure between 90 th
to 95th percentile (z-scores
1.282-1.645), and hypertension >95th
percentile (z-score >1.645), using the sex and height adjusted Centre
for Disease Control (CDC, 2000) standards for the American children
[22]. Dyslipidemia (either
hypertriglyceredemia ≥200
mg/dL, or low HDL cholesterol <40 mg/dL, or high LDL cholesterol
≥100 mg/dL or
hypercholesterolemia ≥170
mg/dL) was defined according to American Heart Association (AHA)
guidelines, endorsed by the American Academy of Pediatrics [23]. At
these cut-offs dietary and/or life style interventions are suggested
[23,24]. Impaired glucose tolerance was defined as fasting plasma
glucose levels ≥100
mg/dL [25]. The AHA cut-off values for fasting plasma glucose and HDL
cholesterol corresponded with the International Diabetes Federation
(IDF) definition of metabolic syndrome in children and adolescents [26].
However, the cut-off for triglyceride was lower (≥1.7
mmol/l or ≥150
mg/dL) with IDF criteria [26].
Statistics: Statistical analysis was done using
SPSS version 13.0 (SPSS Inc, Chicago, Illinois, USA). We had the
recorded age to the exact day, which was grouped into 0.1 yearly
intervals. However, the cut-off values for BMI references were depicted
at half-monthly [5], monthly [4], half-yearly [2] and yearly [17]
intervals. In order to obtain precise numerical cut-offs at 0.1 yearly
intervals, smoothing was required for the intervening time points. This
was achieved by polynomial curve regression [27] using the stated
cut-offs at different ages [2,4,5,17] as anchors, so that these values
remained virtually identical after smoothing. The degree of polynomial
used was based on the best fit and varied for the four references
(seventh degree for the Cole, CDC and WHO references and fourth degree
for IAP reference).
The gender specific age ranges for comparison were
identified according to the ages where the smoothed curves of BMI from
the four references crossed or touched each other (Fig. 1).
Age and sex-specific prevalences of thinness, defined as per the
cut-offs recommended by the four BMI references [2,4,5,17], were
compared. The cut-offs used were below -2 SD (Z score) for WHO [4] and
CDC [5] references; below age and gender specific extrapolation of BMI
of 17 at 18 years, which correspond to a mean Z score of -2, for Cole
et al. [2] reference; and below 5 th
percentile for IAP reference [17].
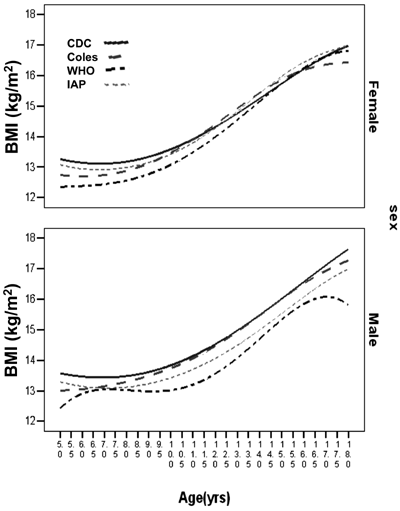 |
|
Fig.1 Comparison of smoothed curves
for body mass index limits for defining thinness.
|
Sex and height adjusted Z-scores for systolic and
diastolic blood pressure of children were calculated using separate
equations as recommended [22].
We also compared the odds ratio, sensitivity,
specificity, predictive values and likelihood ratios for detecting any
cardio-metabolic risk factor in children identified as thin using
various BMI references.
Results
Subjects consenting for blood-letting were more often
females and had significantly greater age, socio-economic class,
height-for-age (0.2 Z-score), weight-for-age (0.8 Z-score) and
BMI-for-age (0.5 Z-score) (P<0.001 for all variables). The age,
gender and socio-economic class adjusted Odds Ratio (95% CI) for being
sampled per unit increase in BMI-for-age Z score was 1.10 (1.09,
1.12).
Descriptive characteristics of the subjects are
summarized in Table I. Overall prevalence of thinness
varied between 6.6% and 16.9% for boys, and 6.5% and 10.3% for girls
using IAP and CDC references, respectively (Tables II and
III). The differences were more pronounced for boys as compared to
girls. Prevalence with IAP reference was substantially lower than CDC
reference for boys across all the age ranges (least for the age range
14.6 to 18.0 years) (Table II). Thinness prevalence in
boys using the CDC and Cole references was 18.2% and 10.7%, respectively
compared to 7.9% and 4.4% using WHO and IAP references for the age range
of 5.0 to 6.5 years (Table II). After this age range,
prevalence with WHO reference was lower than CDC reference but higher
than Cole, et al. [2] reference. Prevalence by Cole, et al.
[2] reference was closer to IAP reference in boys above 6.5 years.
TABLE I Descriptive Characteristics of the Study Population
|
Income group (Number) |
Mean (SD) age (yrs) |
Mean BMI (kg/m2) (SD)[range] |
Sex |
|
|
|
Male
|
Female |
|
LIG (5031) |
12.1 (3.5) |
16.37 (2.7) [11.2 – 32.8] |
2378
|
2653 |
|
MIG (5119) |
11.6 (3.3) |
16.96 (3.1) [11.2 – 36.7] |
2736
|
2383 |
|
HIG (6095) |
11.5 (3.1) |
18.71 (4.5) [11.0 – 39.3] |
3199
|
2896 |
|
Total 16245 |
11.7 (3.3) |
17.44 (3.7) [11.0 – 39.3] |
8313
|
7932 |
|
BMI – Body mass index; HIG – High income group; LIG – Low
income group; MIG – Middle income group. |
TABLE II Prevalence of Thinness in Boys According to Four BMI References
| |
Years |
|
Referencea |
5.0 - 6.5 |
|
Ref |
6.6 - 9.5 |
|
9.6 -12.0 |
|
12.1 -14.5 |
|
14.6 - 18.0 |
|
Ref |
5.0-18.0 |
|
|
n |
% |
|
n |
% |
n |
% |
n |
% |
n |
% |
|
n |
% |
|
CDC |
83 |
18.2 |
CDC |
203 |
11.0 |
233 |
13.4 |
423 |
19.3 |
460 |
22.0 |
CDC |
1402 |
16.9 |
|
Cole |
49 |
10.7 |
WHO |
154 |
8.4 |
211 |
12.2 |
413 |
18.8 |
423 |
20.3 |
WHO |
1237 |
14.9 |
|
WHO |
36 |
7.9 |
Cole |
111 |
6.0 |
127 |
7.3 |
281 |
12.8 |
301 |
14.4 |
Cole |
869 |
10.5 |
|
IAP |
20 |
4.4 |
IAP |
80 |
4.3 |
53 |
3.1 |
175 |
8.0 |
217 |
10.4 |
IAP |
545 |
6.6 |
|
All b |
456 |
|
|
1844 |
|
1733 |
|
2193 |
|
2087 |
|
|
8313 |
|
a The references in each age interval
are ordered according to the cut-off value in descending order;
b total number of subjects in the age range. |
Crossing over of references was more frequent in
girls. In girls, maximum variation was seen in youngest age range
(5.0-9.3 years). Prevalence with IAP reference was consistently lower
compared to other references till the age of 15 years, after which there
were no substantial differences between the references. The WHO cut-off
estimated lower prevalence in girls after the age of 15.6 years;
whereas, CDC cut-off documented lower prevalence after the age of 12.3
years (Table III).
TABLE III Prevalence of Thinness in Girls according to four BMI References
|
Years |
|
5.0-9.3 |
9.4-11.1 |
11.2-12.3 |
12.4-14.1 |
14.2-15.0 |
15.1-15.6 |
15.7-15.8 |
15.9-16.9 |
17.0-18.0 |
5.0-18.0 |
|
Ref a |
n |
% |
Ref |
n |
% |
Ref |
n |
% |
Ref |
n |
% |
Ref |
n |
% |
Ref |
n |
% |
Ref |
n |
% |
Ref |
n |
% |
Ref |
n |
% |
Ref |
n |
% |
|
CDC |
232 |
9.7 |
CDC |
159 |
12.0 |
WHO |
94 |
11.5 |
WHO |
154 |
11.4 |
Cole |
65 |
11.0 |
Cole |
39 |
10.1 |
Cole |
20 |
16.0 |
Cole |
63 |
10.7 |
Cole |
50 |
14.5 |
CDC |
820 |
10.3 |
|
Cole |
160 |
6.7 |
WHO |
134 |
10.1 |
CDC |
85 |
10.4 |
Cole |
146 |
10.8 |
WHO |
62 |
10.5 |
WHO |
36 |
9.4 |
IAP |
15 |
12.0 |
IAP |
53 |
9.0 |
CDC |
49 |
14.2 |
Cole |
749 |
9.4 |
|
WHO |
127 |
5.3 |
Cole |
128 |
9.7 |
Cole |
78 |
9.5 |
CDC |
135 |
10.0 |
CDC |
59 |
9.9 |
IAP |
35 |
9.1 |
WHO |
14 |
11.2 |
CDC |
53 |
9.0 |
IAP |
45 |
13.1 |
WHO |
712 |
9.0 |
|
IAP |
64 |
2.7 |
IAP |
77 |
5.8 |
IAP |
54 |
6.6 |
IAP |
115 |
8.5 |
IAP |
57 |
9.6 |
CDC |
34 |
8.8 |
CDC |
14 |
11.2 |
WHO |
49 |
8.3 |
WHO |
42 |
12.2 |
IAP |
515 |
6.5 |
|
All b |
2401 |
|
All |
1324 |
|
All |
819 |
|
All |
1352 |
|
All |
593 |
|
All |
385 |
|
All |
125 |
|
All |
589 |
|
All |
344 |
|
|
7932 |
|
a The references in each age interval are ordered
according to the cut-off value in descending order.
b total number of subjects in the age range. |
Children identified as thin by any reference had a
significantly lower risk of associated cardio-metabolic risk factors (Table
IV); IAP cut-off had the least odds ratio (0.59) but this was
not significantly different from the other three references (overlapping
confidence intervals). Among children with pre-hypertension,
hypertension or any cardio-metabolic aberration, the prevalence of
thinness was highest with CDC and least with IAP definition (Table
V; P<0.01). These differences were greater in boys (2.5
to 3 fold) than in girls (1.3 to 2 fold). In boys the thinness
prevalence was greater with WHO in comparison to Cole reference, but the
converse was true for girls.
TABLE IV Comparison of Four Thinness cut-offs for Diagnosing any Cardio-metabolic Aberration
|
Thinness |
Odds ratio (95%
|
Sensitivity |
Specificity |
Positive |
Negative |
Positive |
Negative
|
|
cut-off |
CI; P value |
|
|
predictive
|
predictive
|
likelihood
|
likelihood
|
|
|
|
|
value |
value |
ratio |
ratio
|
|
CDC |
0.73 (0.57, 0.93); 0.011 |
0.10 |
0.87 |
0.60 |
0.32 |
0.75 |
1.04 |
|
WHO |
0.73 (0.56, 0.93); 0.012 |
0.09 |
0.88 |
0.60 |
0.32 |
0.75 |
1.03 |
|
Cole |
0.62 (0.48, 0.81); 0.001 |
0.07 |
0.89 |
0.57 |
0.32 |
0.65 |
1.04 |
|
IAP |
0.59 (0.43, 0.81); 0.001 |
0.05 |
0.92 |
0.55 |
0.32 |
0.61 |
1.03 |
Any cardio-metabolic aberration was defined as presence of one or more of the following in an individual subject: fasting levels of blood glucose
≥100 mg/dl or total cholesterol
≥170 mg/dl or LDL cholesterol
≥100 mg/dl or HDL cholesterol <40 mg/dl or serum triglyceride
≥200 mg/dl.
Odds Ratios were computed by logistic regression analysis and pertain to comparison of thin with non-thin subjects. |
TABLE V Prevalence of Thinness according to four Definitions in Children with Pre-hypertension, Hypertension or any Cardio-metabolic Aberration
| |
Boys |
|
Girls |
|
Definition |
Prehypertension |
Hypertension |
Any cardio- |
Definition |
Prehypertension |
Hypertension |
Any cardio- |
|
(1429 of 8313) |
(2745 of 8313) |
metabolic aberration |
|
(1310 of 7932) |
(2875 of 7932) |
metabolic aberration |
|
|
|
(906 out of 1320) |
|
|
|
(967 out of 1476) |
|
Thinness prev (%) |
Thinness prev(%) |
Thinness prev (%) |
|
Thinness prev (%) |
Thinness prev(%) |
Thinness prev (%)
|
|
(n of 1429) |
(n of 2745) |
(n of 906) |
|
(n of 1310) |
(n of 2875) |
(n of 967) |
|
CDC |
15.9 (227) |
10.1 (276) |
11.8 (107) |
CDC |
10.8 (141) |
6.9 (198) |
7.5 (73) |
|
WHO |
13.7 (196) |
8.2 (225) |
11.0 (100) |
Cole |
8.6 (112) |
6.0 (172) |
7.4 (72) |
|
Cole |
10.2 (146) |
5.7 (156) |
6.7 (61) |
WHO |
8.1 (106) |
5.6 (161) |
7.9 (76) |
|
IAP |
6.4 (92) |
3.4 (94) |
4.0 (36) |
IAP |
5.9 (77) |
3.5 (101) |
5.6 (54) |
Prev – Prevalence
Any cardio-metabolic aberration was defined as presence of one or more of the following in an
individual subject: fasting levels of blood glucose ³100 mg/dL or total cholesterol
≥170 mg/dl or LDL cholesterol ≥100 mg/dl or HDL cholesterol <40 mg/dL or serum triglyceride ≥200 mg/dL.
|
Discussion
The prevalence of thinness varied with the reference
employed, especially in boys. In comparison to IAP reference, CDC
reference overestimated thinness maximally (2-4 fold in boys and up to
3.5 fold in girls). This overestimation was lower with WHO and Cole
references. Children identified as thin by any reference had comparable,
significantly lower risks of associated cardio-metabolic aberrations.
However, in subjects with any cardio-metabolic or blood pressure
aberration, the prevalence of thinness was highest with CDC and least
with IAP definition.
We have evaluated a large sample of urban school
children from different socio-economic strata in a developing country
undergoing nutrition transition [13]. Simultaneous differences in the
prevalences of anthropometric thinness and cardio-metabolic aberrations
warranting interventions [23-26] were quantified. Serum biochemistry was
performed in an international quality controlled laboratory.
The following limitations merit consideration: (i)
We could not adjust for the influence of pubertal growth spurts on BMI
as sexual maturity staging was not done. However, this has little
relevance as the four evaluated references do not recommend such
adjustment for quantifying thinness; (ii) In comparison to other
criteria, IAP reference [17] had an uncharacteristic shape for boys,
especially above 16 years (Fig. 1). This is not due to our
smoothing for intervening age-periods but probably reflects weaknesses
in original IAP reference – small sample sizes at these ages and not
using the recommended LMS method for smoothing. Nevertheless, this has
no implications for our stated objectives, because thinness will be
categorized according to the depicted cut-offs; (iii) Blood
sampled participants had significant differences from others including a
higher BMI. This has implications for determining overall prevalence of
cardio-metabolic risk factors but not for evaluating their relationship
with thinness; (iv) The high prevalence of pre-hypertension and
hypertension needs cautious interpretation. We used CDC percentiles for
defining height adjusted pre-hypertension and hypertension rather than
an Indian reference to maintain uniformity with other biochemical
cut-offs. In comparison to unadjusted estimates as in many earlier
studies, height adjustment lowers the reference BP thereby inflating the
pre-hypertension and hypertension prevalence in shorter populations as
in New Delhi (mean height SD score -0.73). We also acknowledge the
inherent challenges of blood pressure measurements in children [28].
Blood pressure was recorded during one visit only, which may not reflect
sustained hypertension. Among 15-16 year old students from Dallas, USA
with elevated BP (≥95 th
percentile) at initial examination, only 17% had sustained elevated BP
over 2 subsequent visits in the same year [29]. Other studies also
indicate that the majority of children with elevated BP at a given visit
have normal BP at subsequent visits a few weeks later, notably because
of regression to the mean and habituation to the measurement procedure
[28]. It would thus have been ideal for us to perform two repeat BP
measurements; however, paucity of resources precluded this possibility.
A high prevalence of systolic or diastolic incident hypertension in
children (17.3% in overweight vs. 10.1% in others) has been shown
in a study on a mixed rural and urban Southern Indian population [30].
Thus the overestimation of blood pressure from a single visit in our
data from urban area may not be substantial; and (v). A larger
sample size would have provided greater power to detect the observed
differences in predictive abilities of the references for identifying
absence of any cardio-metabolic aberration.
We are unaware of any similar studies comparing the
prevalences of cardio-metabolic aberrations in thin children. However,
anthropometric and metabolic discordance has been documented in
"metabolically obese normal weight" children in a nationally
representative sample from Iran [31]. Several studies have documented
variations in prevalences of obesity and metabolic syndrome in children
and adolescents with use of different references [6-11]. In younger
children, overestimation of prevalence of undernutrition with CDC
reference has been documented in both developing and developed countries
[32-35]. We used cut-off
criteria for which nutritional and/or lifestyle interventions are
suggested by the AHA [22-25].
The use of IDF definition
[26] increased the prevalence of hyper-triglyceridemia
from 2.1% to 10.5% without much difference for any cardio-metabolic
aberration (67% vs. 68%).
From a clinician’s perspective, labeling of an
individual child as "thin" leads to further diagnostic and therapeutic
actions. Absence of data comparing the appropriateness of the management
interventions with various cut-offs leads to uncertainty on the utility
of one BMI reference over other. Routine recording of blood pressure
after the age of three years is recommended in developed country
settings to counter obesity related hypertension [24]. It may be a good
practice for pediatricians in India to replicate this guideline
irrespective of BMI status. Similarly, there may be a case for
evaluating the metabolic profile in "at risk" thin children as
identified by high blood pressure or a parental history of premature
coronary artery disease, hypertension, diabetes mellitus or dyslipidemia.
Future research on the utility of these recommendations is required.
In a public health setting, two-to four-fold relative
variations in prevalences of thinness have socio-political implications
for ranking the development status and progress of nations. Unlike the
WHO growth references for under-five children that are conceptualized on
prescriptive practices, these evaluated BMI cut-offs are based on
statistical charts, which have not been linked to important biological
or functional consequences [36,37].
The usual public health and clinical response in thin
children is to recommend food supplementation [38,39], which has the
potential to worsen cardio-metabolic risk factors including blood
pressure. Children identified as thin by any reference had a
significantly lower risk of cardio-metabolic aberration (positive
likelihood ratios of 0.61 to 0.75 for detecting cardio-metabolic
aberration). Opinions will vary on the usefulness of these predictions.
However, according to conventional guidelines [40], such likelihood
ratios of <0.2 are generally considered important clinically while
values >0.5 are labeled worthless.
Biomarkers can be considered as more proximate
reflectors of recent nutritional balance; thus cardio-metabolic
aberrations reflect metabolic overnutrition, even in anthropometrically
thin ("undernourished") subjects. In this context, from a public health
perspective, national reference has the advantage of detecting the least
prevalence of thinness in metabolically overnourished individuals.
However, further research is needed to validate these findings in
different settings.
In conclusion, prevalences of thinness in
school-children vary considerably with the reference employed,
especially in boys. Thin children, identified by any reference, have a
lower risk of associated cardio-metabolic aberrations; however, thinness
is a poor diagnostic test for this purpose. In populations undergoing
nutrition transition, there is a need to link cardio-metabolic risk
factors with recommended anthropometric criteria to define
undernutrition.
Acknowledgements: We thank Lieutenant Colonel Dr.
Rajat Prakash for training nutritionists, Supreet Kaur (SK), Lakshmi
Labani (LL) and Shikha Shukla (SS) for anthropometric and blood pressure
measurements.
Contributors: HPSS, UK and PG: conceived the
study, analysis and co-drafted the manuscript; CO, HPSS, DG, SS and PG:
were responsible for data management and analysis; UK and SK: were
responsible for the field work including blood sampling and
anthropometry; RL: was responsible for laboratory analyses and their
quality control; All the authors contributed to and approved the
submitted version; HPSS and UK: will act as co-guarantors.
Funding: Indian Council of Medical
Research (ICMR); Competing interests: None stated.
References
1. Bailey KV, Ferro-Luzi A. Use of body mass index of
adults in assessing individual and community nutritional status. Bull
WHO. 1995;73:673-80.
2. Cole TJ, Flegal KM, Nicholls D, Jackson AA. Body
mass index cut offs to define thinness in children and adolescents:
international survey. BMJ. 2007;335:194-201.
3. Cameron N. Body mass index cut offs to define
thinness in children and adolescents. BMJ. 2007;335:166-7.
4. World Health Organization. Growth reference 5-19
years: BMI-for-age (5-19 years). Available at
http://www.who.int/growthref/who2007_bmi_for_age/en/index.html (Accessed
December 28, 2012).
5. Centers for Disease Control and Prevention. Growth
charts: Z score data files. Available at
http://www.cdc.gov/growthcharts/zscore.htm (Accessed December 28, 2012).
6. Al-Sendi AM, Shetty P, Musaiger AO. Prevalence of
overweight and obesity among Bahraini adolescents: a comparison between
different sets of criteria. Eur J Clin Nutr. 2003;57:471–4.
7. O’Neill JL, McCarthy SN, Burke SJ, Hannon EM,
Kiely M, Flynn A, et al. Prevalence of overweight and obesity in
Irish school children, using four different definitions. Eur J Clin Nutr.
2007;61:743-51.
8. Ko GT, Ozaki R, Wong GW, Kong AP, So WY, Tong PC,
et al. The problem of obesity among adolescents in Hong Kong: a
comparison using various diagnostic criteria. BMC Pediatr. 2008;8:10.
9. Lee S, Bacha F, Gungor N, Arslanian S. Comparison
of different definitions of pediatric metabolic syndrome. Relation to
abdominal adiposity, insulin resistance, adinopectin, and inflammatory
biomarkers. J Pediatr. 2008;152:177-84.
10. Ford ES, Li C. Defining the metabolic syndrome in
children and adolescents: Will the real definition please stand up? J
Pediatr. 2008;152:160-4.
11. Reinehr T, de Sousa G, Toschke AM, Andler W.
Comparison of metabolic syndrome prevalence using eight different
definitions: a critical approach. Arch Dis Child. 2007;92:1067-72.
12. National Family Health Survey-3. Available
http://www.nfhsindia.org/volume_1.html (Accessed 1st April 2008).
13. Shetty PS. Nutrition transition in India. Public
Health Nutr. 2002;5:175-82.
14. Sawaya AL, Sesso R, Florêncio TM, Fernandes MT,
Martins PA. Association between chronic undernutrition and hypertension.
Matern Child Nutr. 2005;1:155-63.
15. Stein AD, Thompson AM, Waters A. Childhood growth
and chronic disease: evidence from countries undergoing the nutrition
transition. Matern Child Nutr. 2005;1:177-84.
16. Victora CG, Adair L, Fall C, Hallal PC, Martorell
R, Richter L, et al. Maternal and child under-nutrition:
consequences for adult and human capital. Lancet.
2008;371:340-57.
17. Khadilkar VV, Khadilkar AV, Choudhury P, Agarwal
KN, Ugra D, Shah NK. IAP growth monitoring guidelines for children from
birth to 18 years. Indian Pediatr. 2007;44:187-97.
18. Physical status: the use and interpretation of
anthropometry: report of a WHO expert committee. World Health Organ Tec
Rep Ser. 1995;854:329.
19. Lolekha PH, Srisawasdi P, Jearanaikoon P,
Wetprasit N, Sriwanthana B, Kroll MH. Performance of four sources of
cholesterol oxidase for serum cholesterol determination by the enzymatic
end point method. Clinica Chimica Acta. 2004;339:135-45.
20. Henkel E, Stoltz M. A newly drafted color test
for the determination of triglycerides convenient for manual and
mechanized analysis (glycerolphosphate-oxidase-PAP). Fresenius J Anal
Chem. 1982;311:451-2.
21. Friedewald WT, Levy RI, Fredrickson DS.
Estimation of the concentration of low-density lipoprotein cholesterol
in plasma, without use of the preparative ultracentrifuge. Clin Chem.
1972;18:499-502.
22. National High Blood Pressure Education Program
Working Group on High Blood Pressure in Children and Adolescents. The
Fourth Report on the Diagnosis, Evaluation, and Treatment of High Blood
Pressure in Children and Adolescents. Pediatrics.
2004;114:555-76.
23. Gidding SS, Dennison BA, Birch LL, Daniels SR,
Gillman MW, Lichtenstein AH, et al. Dietary recommendations for
Children and Adolescents: A guide for Practitioners: Consensus Statement
from the American Heart Association. Circulation. 2005;112:2061-75.
24. Williams CL, Hayman LL, Daneils SR, Robinson TN,
Steinberger J, Paridon S, et al. Cardiovascular Health in
Childhood: A statement for health professionals from the Committee on
Atherosclerosis, Hypertension, and Obesity in the Young (AHOY) of the
council on cardiovascular disease in the young, American Heart
Association. Circulation. 2002;106:143-60.
25. Kavey RE, Allada V, Daniels SR, Hayman LL,
McCrindle BW, Newburger JW, et al. Cardiovascular risk reduction
in high-risk pediatric patients: A scientific statement from the
American Heart Association (AHA) expert panel on population and
prevention science; the councils on cardiovascular disease in the young,
epidemiology and prevention, nutrition, physical activity and
metabolism, high blood pressure research, cardiovascular nursing, and
the kidney in heart disease; and the Interdisciplinary working group on
quality of care and outcomes research. Circulation. 2006;114:2710-38.
26. Zimmet P, Alberti G, Kaufman F, Tajima N, Silink
M, Arslanian S, et al. The metabolic syndrome in children and
adolescents. Lancet. 2007;369:2059-61.
27. Zar JH. Polynomial regression. In: Zar JH,
editors. Biostatistical Analysis. 4th ed. New Jersey: Prentice Hall;
1999. p. 452-7.
28. Chiolero A, Bovet P, Paradis G. Screening for
elevated blood pressure in children and adolescents. A critical
appraisal. JAMA Pediatr. Published online January 7, 2013.
doi:10.1001/jamapediatrics.2013.438.
29. Fixler DE, Laird WP. Validity of mass blood
pressure screening in children. Pediatrics. 1983;72:459-63.
30. Raj M, Sundaram KR, Paul M, Deepa AS, Kumar KR.
Obesity in Indian children: Time trends and relationship with
hypertension. Natl Med J India. 2007;20:288-93.
31. Kelishadi R, Cook SR, Motlagh ME, Gouya MM,
Ardalan G, Motaghian M, et al. Metabolically obese normal weight
and phenotypically obese metabolically normal youths: the CASPIAN study.
J Am Diet Assoc. 2008;108:82-90.
32. de Onis M, Onyango AW, Borghi E, Garza C, Yang H.
Comparison of the World Health Organization child growth standards and
the National Centre for Health Statistics/WHO international growth
reference: implications for child health programmes. Public Health Nutr.
2006;9:942-7.
33. Deshmukh PR, Dongre AR, Gupta SS, Garg BS. Newly
developed WHO standards: implications for demographic surveys and child
health programs. Indian J Pediatr. 2007;74:987-90.
34. Roberfroid D, Lerude MP, Pérez-Cueto A, Kolsteren
P. Is the CDC 2000 growth reference appropriate for developing
countries? Public Health Nutr. 2006;9:266-8.
35. Kelishadi R, Ardalan G, Gheiratmand R, Majdzadeh
R, Hosseini M, Gouya MM, et al. Thinness, overweight and obesity
in a national sample of Iranian children and adolescents: CASPIAN study.
Child Care Health Dev. 2008;34:44-54.
36. Seidell JC, Doak CM, de Munter JS, Kuijper LD,
Zonneveld C. Cross sectional growth references and implications for the
development of an international growth standard for school-aged children
and adolescents. Food Nutr Bull. 2006;27:S189-98.
37. Himes JH. Long term longitudinal studies and
implications for the development of an international growth reference
for children and adolescents. Food Nutr Bull. 2006;27: S199-211.
38. Greenhalgh T, Kristjansson E, Robinson V.
Realistic review to understand the efficacy of school feeding
programmes. BMJ 2007; 335: 858-61.
39. Kristjansson EA, Robinson V, Pettiscrew M,
MacDonald B, Krasevac J, Janzel L, et al. School feeding for
improving the physical and psychosocial health of disadvantaged
elementary school children. Cochrane Database Syst Rev 2000;
24:CD004676.
40. Jaeschke R, Guyatt G, Lijmer J. Diagnostic tests.
In: Users’ Guides to the Medical Literature. Essentials of
Evidence Based Clinical Practice. The Evidence-Based Medicine Working
Group. Eds. Guyatt G, Rennie D. JAMA and Archives Journals,
American Medical Association Press, 2004; p. 187-217.
|
|
|
 |
|

