Acute lymphoblastic leukemia
(ALL) is one of
the most common haematological
malignancies of childhood treated with high
dose methotrexate not only to prevent central nervous system (CNS)
recurrence but also hematologic relapses. The association between
methotrexate therapy and idiosyncratic neurological complications is
well documented [1] but intracranial hemorrhage following intrathecal
methotrexate administration is extremely rare. We report a 2½-year-old
girl who developed both intraventricular and subarachnoid hemorrhage
following intrathecal methotrexate administration.
Case Report
A 2½-year-old girl presented with a 2 week history of
fever, progressive pallor, petechial spots, hepato-splenomegaly and
generalized lymphadenopathy. She was diagnosed as ALL Type L1 (FAB
classification); CNS involvement was excluded by cerebrospinal fluid
(CSF) analysis in the pre-treatment period. She was categorized as
standard risk group and was then put to induction phase 1A of
chemotherapy with prednisolone, vincristine, daunorubicine,
L-asparaginase and intrathecal methotrexate.
Following completion of induction phase 1A, patient
showed initial remission as evident by repeat bone marrow examination.
Phase 1B of induction therapy was then started with 6-Mercaptopurine,
cylophosphamide, cytarabine and intrathecal methotrexate. On 3rd day
after receiving last dose of intrathecal methotrexate, patient developed
vomiting, sudden onset generalized tonic-clonic seizure and alteration
of consciousness. Blood pressure was measured as 112/78 mmHg. There was
no history of trauma or any leak of CSF from the lumbar puncture site.
Patient was shifted to pediatric intensive care unit. Complete blood
count (CBC) revealed hemoglobin 9.7 g/dL, white blood cell count 3.6×10
3/µL
and platelet count 140×103/µL.
Coagulation profile showed normal prothrombin time and activated partial
thromboplastin time. Non-contrast axial computed tomography (CT) scan of
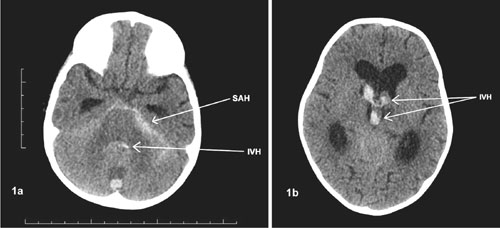
brain showed ventricular and subarachnoid hemorrhage (Fig. 1).
 |
|
Fig. 1 Non-contrast axial CT scan of
brain showing (a) subarachnoid haemorrhage in prepontine
cisterns and hemorrhage within 4th ventricle; and (b)
haemorrhage in 3rd and both lateral ventricles.
|
Patient was treated conservatively by adequately
controlling raised intracranial tension, seizures and providing constant
supportive and nursing care. Patient regained consciousness and was able
to take oral feeds. MRI of brain after four weeks showed revealed
resolution of hemorrhage with mildly dilated ventricles as a sequel.
Magnetic resonance angiography (MRA) could not demonstrate any
underlying vascular malformation. Presently, she is receiving
chemotherapy as per the above mentioned protocol.
Discussion
Intracerebral hemorrhage (ICH) following intrathecal
methotrexate administration is an extremely rare but serious life
threatening complication. A few cases have been reported in the
literature till date [2-4]. As the other possible etiologies of ICH like
arteriovenous mal-formation, hypertension, thrombocytopenia,
coagulo-pathy, history of major head trauma or thrombolytic therapy were
excluded in the present case, intrathecal methotrexate was considered to
be the probable cause in our patients.
The exact pathogenesis for intracranial bleeding
following intrathecal administration of methotrexate is unknown. Faulty
technique during lumbar puncture can cause significant CSF leakage
resulting in low intracranial pressure leading to traction and rupture
of the dilated, thin-walled dural blood vessels [5-7].
In our case, there was no such history of CSF leak.
Intraventricular and subarachnoid hemorrhage in our case occurred
possibly due to vasculopathic effect of methotrexate in the central
nervous system. The vasculopathic effect of methotrexate may be due to
fibrinoid degeneration and hyaline thrombus of the cerebral vessels as
described earlier [8, 9]. Alteration in the regional cerebral blood flow
could be another pathogenic mechanism for ICH [10].
Intrathecal methotrexate is routinely used for
prevention of CNS involvement in patients with ALL. Mild and transient
neurological complications following IT methotrexate are common but
intraventricular and subarachnoid hemorrhage may rarely occur.
Clinicians should be aware of this rare but serious adverse effect of
intrathecal methotrexate.
Contributors: RP, TKD: diagnosed, worked up the
case and wrote the manuscript; BD, CL: managed the case and reviewed the
literature. RP, TKD: prepared the final manuscript and followed up the
case.
Funding: None; Competing interest: None
stated.
References
1. Salkade PR, Lim TA. Methotrexate-induced acute
toxic leukoencephalopathy. J Cancer Res Ther. 2012;8:292-6.
2. Lakhkar BN, Sinha R. Intrathecal methotrexate
therapy complication. Indian J Radiol Imaging. 1999;9:31.
3. Alexopoulou A, Dourakis SP, Georgousi KK,
Archi-mandritis AJ. Intracerebral hematoma following intrathecal
administration of methotrexate in a patient with non-Hodgkin’s lymphoma.
Am J Hematol. 2005;78:159-60.
4. Boran P, Tokuc G, Boran BO, Oktem S. Intracerebral
hematoma as a complication of intrathecal methotrexate administration.
Pediatr Blood Cancer. 2008;50:152-4.
5. Vos PE, de Boer WA, Wurzer JA, van Gijn J.
Subdural hematoma after lumbar puncture: Two case reports and review of
the literature. Clin Neurol Neurosurg. 1991;93:127-32.
6. Gupta SR, Naheedy MH, Rubino FA. Cranial subdural
hematoma following lumbar myelography. Comput Radiol. 1985;9:129-31.
7. Van de Kelft E, Bosmans J, Parizel PM, Van Vyve M,
Selosse P. Intracerebral hemorrhage after lumbar myelography with
iohexol: Report of a case and review of the literature. Neurosurgery.
1991;28:570-4.
8. Shapiro WR, Chernik NL, Posner JB. Necrotizing
encephalopathy following intraventricular instillation of methotrexate.
Arch Neurol. 1973;28:96-102.
9. Greenhouse A, Neuberger KT, Bowerman DL. Brain
damage after intracarotid infusion of methotrexate. Arch Neurol.
1964;11:618-22.
10. Osterlundh G, Bjure J, Lannering B, Kjellmer I,
Uvebrant P, Márky I. Studies of cerebral blood flow in children with
acute lymphoblastic leukemia: Case reports of six children treated with
methotrexate examined by single photon emission computed tomography. J
Pediatr Hematol Oncol. 1997;19:28-34.

