|
|
|
Indian Pediatr 2014;51:
375-378 |
 |
Zinc Supplementation for Neonatal
Hyperbilirubinemia: A Randomized Controlled Trial
|
|
Ashok Kumar, Narendra Kumar Bagri, Sriparna Basu and *Ravi Kumar
Asthana
From Departments of Pediatrics and *Botany, Banaras Hindu University,
Varanasi, India
Correspondence to: Dr Ashok Kumar, Neonatal Unit, Department of
Pediatrics, Institute of Medical Sciences, Banaras Hindu University,
Varanasi 221 005, India.
Email:
[email protected]
Received: August 28, 2013;
Initial review: September 24, 2013;
Accepted: March 12, 2014.
|
Objectives: To determine the efficacy of oral zinc for treatment of
idiopathic neonatal hyperbilirubinemia in near-term and term (35-41
weeks) neonates.
Design: Randomized placebo-controlled trial.
Setting: Tertiary-care teaching hospital
Participants: Eighty newborns with idiopathic
neonatal hyperbilirubinemia.
Intervention: Neonates were randomized to receive
either oral zinc sulfate (10 mg/d) or placebo for 7 days.
Main outcome measures: Primary: total serum
bilirubin levels at 48 (±12) h, 96 (±12) h and 144 (±12) h after
intervention. Secondary: duration of phototherapy, and serum zinc and
copper levels.
Results: Baseline mean (SD) total serum bilirubin
levels were 14.8 (3.8) and 14.4 (3.5) mg/dL in zinc and placebo groups,
respectively. No significant differences were observed in total
bilirubin levels between the two groups after the intervention. Mean
(SD) total serum bilirubin levels in zinc and placebo groups were 13.9
(2.5) vs. 13.4 (1.9) mg/dL (mean difference 0.566; 95% CI -0.535,
1.668, P=0.038) at 48 h, 13.1 (2.7) vs. 12.8 (2.3) mg/dL
(mean difference 0.234; 95% CI -1.011, 1.479, P =0.708) at 96 h
and 8.0 (2.0) vs. 8.6 (1.2) mg/dL (mean difference -0.569, 95% CI
-1.382, 0.242, P=0.166) at 144 h. Although the mean duration of
phototherapy in the zinc group was less by 21.3 h (95% CI 11.6, 30.9,
P=0.052), the difference was not significant. Post-intervention,
serum zinc levels were significantly higher in the zinc-supplemented
group while serum copper levels were comparable between the two groups.
Conclusions: Oral zinc sulfate, in a dose of
10 mg/day, is not effective in the management of idiopathic neonatal
hyperbilirubinemia.
Keywords: Jaundice, Neonate, Zinc compounds
|
|
S
tandard treatment for neonatal hyperbilirubinemia
consists of phototherapy and/or exchange transfusion depending on serum
bilirubin levels. Although effective, both these modalities necessitate
hospital admission, increase the cost of care, expose the baby to the
risk of infection, and are associated with side effects [1,2].
Enterohepatic circulation (EHC) of bilirubin contributes significantly
to neonatal hyperbilirubinemia [3], and its blockage might be a
therapeutic target. Oral zinc salts have been shown to decrease serum
bilirubin levels in hyperbilirubinemic rats, presumably by inhibition of
EHC [4]. However, the role of zinc in disrupting EHC in human neonates
is not clear. A recent trial has failed to demonstrate any beneficial
effect of zinc on the incidence of hyperbilirubinemia in at-risk
neonates [5].
This randomized, placebo-controlled trial was
conducted to determine the effect of oral zinc supplementation on total
serum bilirubin (TSB) levels in idiopathic neonatal hyperbilirubinemia.
Methods
The study was conducted in Sir Sunderlal Hospital,
Banaras Hindu University, Varanasi, India from February 2008 to May
2009. It was approved by the Institute Ethics Committee. Study
population included term (37-41 weeks) and near-term (35-36 weeks)
newborns with idiopathic neonatal hyperbilirubinemia. The inclusion
criteria were: (i) appearance of jaundice after 24 hours of age;
(ii) total serum bilirubin (TSB) exceeding the 40th percentile
track for age as per the hour-specific bilirubin nomogram of the
American Academy of Pediatrics (AAP) [6]; and (iii) absence of
any obvious cause of neonatal hyperbilirubinemia. Exclusion criteria of
the study were: (i) presence of any systemic illness, and (ii)
major congenital malformations. A written informed consent was taken
from all parents before inclusion of their child in the study.
The bottles of zinc sulfate (10 mg/mL; 10mL) and
placebo were procured from Apex Pharmaceuticals Private Limited,
Chennai, India. Bottles and contents for both the groups were identical
in color and appearance. Information about the content of bottles was
supplied in separate sealed envelope that was not opened until the end
of study. Randomization was done using random numbers table. These
numbers were written on the bottles, taking one bottle from each box,
with each block consisting of 6 or 8.
Following investigations were done to exclude known
cause of jaundice: blood group of the mother and baby, hemoglobin,
complete blood count, reticulocyte count, peripheral blood smear
examination, Coomb’s test, free T 4
and thyroid stimulating hormone (TSH), and G6PD assay. Neonates were
managed as per AAP clinical practice guidelines [7]. The infants were
given 0.5 mL of the solution (either zinc sulfate or placebo) - twice
daily by the mother - for 7 days, using a calibrated dropper provided
with the bottle. TSB levels were estimated at 48 (12) h, 96 (12) h and
144 (12) h, after starting the intervention, or more frequently, if
indicated. The total duration of phototherapy was noted.
Any adverse effect like retching, vomiting, abdominal
distension, diarrhea, skin rash, irritability/crying, change in infant’s
behavior or physical signs were noted by parents and/or physicians.
Bottles were checked for residual content to ensure proper utilization.
Serum zinc and copper levels were estimated using atomic absorption
spectrophotometry (Perkin Elmer, USA, Model No. 2380) in infant’s blood
at baseline and at the end of intervention.
The primary outcome variables were TSB levels at 48
(±12) h, 96 (±12) h and 144 (±12) h after starting the intervention.
Secondary outcome variables were the duration of phototherapy, and serum
zinc and copper levels at the end of intervention.
With a probability of type I error ( a)
of 0.05 and a power of 0.90, a minimum sample size of 37 newborns in
each group was calculated to detect a reduction of at least 3 mg/dL in
the mean TSB levels in the zinc supplemented group. To account for loss
to follow-up, a total of 40 newborns were enrolled in each group.
Statistical analysis: Data were entered in
Microsoft excel software and analyzed by SPSS version 16.0. Parametric
data were expressed as percentage, mean, median and inter quartile
range. Student’s t-test was used to analyze continuous variables.
Chi-squared test was used to test associations between all categorical
variables and Fisher exact probability test was applied to compare
proportions. To compare multiple means, ANOVA test was used and 95%
confidence intervals (CI) were calculated. A P value <0.05 was
taken as statistically significant.
Results
Eighty term and near-term babies were randomly
allocated to receive zinc (n=40) or placebo (n=40).
Fig. 1 shows the flow of participants in the study. Both the
groups were comparable for birth weight, gestational age, proportion of
low birth weight newborns, gender, mode of feeding, weight loss, age of
intervention and laboratory parameters. The baseline serum bilirubin,
serum zinc and copper levels were also similar between placebo and zinc
groups (Table I).
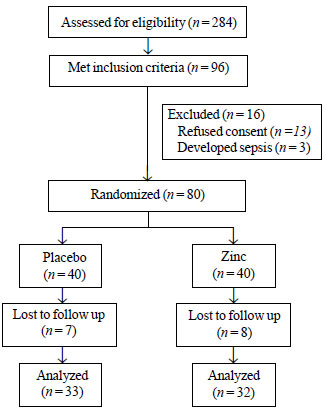 |
|
Fig. 1 Flow of participants in the
study.
|
TABLE I Baseline Characteristics of Study Population
|
Parameters
|
Zinc
|
Placebo
|
|
(n=40) |
(n=40) |
|
Maternal age, y (Mean±SD) |
25.2 ± 4.1 |
26.4±3.8 |
|
History of jaundice in sibling, n(%) |
4 (10) |
2(5.0) |
|
Parity, median |
1 |
1 |
|
Cesarean delivery, n (%) |
24(60.0) |
20(50.0)
|
|
Gestational age (wks) (Mean ± SD) |
37.6±1.5 |
37.7±1.4 |
|
Near terms (35-36 wks), n (%) |
7(17.6) |
5(12.5) |
|
Birth weight (g) (Mean ± SD) |
2711±475 |
2672±400 |
|
Low birth weight (<2500g), n (%) |
14(35.0) |
15(37.5) |
|
Weight loss >10%, n (%) |
4(10) |
3(7.5) |
|
Weight at enrolment (g) (Mean ± SD) |
2595±427 |
2554±416 |
|
Males, n (%) |
28(70.0) |
25(62.5) |
|
Exclusively breast fed, n (%) |
36(90.0) |
34(85.0) |
|
Hematocrit (Mean ± SD) |
48.3±5.1 |
47.5±4.3 |
|
Age (h) at enrolment, Median (IQR) |
82.4(52,132) |
78.5 (54,125) |
|
Serum zinc levels (mg/dL) |
54.8±5.9 |
48.4±6.5 |
|
Serum copper levels (mg/dL) |
60.5±16.3 |
54.4±15.8 |
|
Serum bilirubin levels, mg/dL, (Mean ± SD) |
13.9±2.5 |
13.4±1.9 |
After intervention, no difference in TSB levels was
observed between placebo and zinc groups anytime during the study
period. The duration of phototherapy was 21.3 h less in zinc group in
comparison with placebo but the difference did not reach statistical
significance (Table II). None of the newborns in the two
groups had a TSB level exceeding 20 mg/dL and none required exchange
transfusion. At the end of intervention, serum zinc levels were
significantly higher in the zinc supplemented group while serum copper
levels were comparable between the two groups. The adverse events were
comparable between the two groups (vomiting 3 vs. 2; skin rash 3
vs. 3; diarrhea 4 vs. 3; excessive cry 2 vs. 2 in
placebo and zinc group, respectively).
TABLE II Comparison of Outcome Variables in Study Population
|
Zinc (n=40) |
Placebo (n=40) |
Mean difference (95% CI) |
P value |
|
#TSB levels at 48 ± 12 h, Mean ± SD |
13.9 ± 2.5 |
13.4 ± 1.9 |
0.566 (-0.535, 1.668) |
0.308 |
|
#TSB levels at 96 ± 12 h, Mean ± SD |
13.1 ± 2.7 |
12.8 ± 2.3 |
0.234 (-1.011, 1.479) |
0.708 |
|
#TSB levels at 144 ± 12 h, Mean ± SD |
8.0 ± 2.0 |
8.6 ± 1.2 |
-0.569 (-1.382, 0.242) |
0.166 |
|
Phototherapy given, n (%) |
21 (52.0) |
18 (45.0) |
0.736* (0.282, 1.921) |
0.629 |
|
Age at starting phototherapy, h, Median (IQR) |
87(68, 104) |
92 (81, 105) |
-2.46 (-16.950, 12.029) |
0.733 |
|
Duration of phototherapy, h, Mean ± SD |
61.9 ± 12.1 |
83.3 ± 17.6 |
-21.292 (-30.954, -11.629)
|
0.052 |
|
#Post-intervention serum zinc levels (mg/dL) |
72.2 ± 25.7 |
47.8 ± 17.7 |
24.394 (13.475, 35.312) |
0.000 |
|
#Post-intervention serum copper levels (mg/dL) |
46.2 ± 16.0 |
48.6 ± 15.2 |
-2.468 (-10.214, 5.279) |
0.527 |
|
TSB - Total serum bilirubin (mg/dL); * Odds ratio; #
n=32 for zinc and n=33 for placebo. |
Discussion
The present study showed that there is no beneficial
effect of oral zinc in neonatal hyperbilirubinemia. TSB levels were
comparable in the two groups during the intervention period. Although
the mean duration of phototherapy in the zinc group was less by 21.3h,
the difference was not significant. We included only idiopathic cases of
neonatal hyperbilirubinemia where entero-hepatic circulation plays a
dominant role in the causation of jaundice, and excluded newborns who
had jaundice due to bilirubin overproduction such as ABO/Rh
incompatibility, and G6PD deficiency.
The limitations of the study were small sample size
and loss to follow-up. Moreover, we did not evaluate the impact of
UGT1A1 gene variants on bilirubin load. In a recent study by Rana,
et al. [5], the incidence of hyperbilirubinemia was comparable in
both the zinc and placebo groups (17.9% vs 19.1%). The
requirement of phototherapy was also comparable; but the duration of
phototherapy was shorter in zinc group. However, the above study
included newborns with ABO incompatibility which might have influenced
the results. ABO incompatibility produces hyperbilirubinemia by
hemolysis, and zinc works by blocking entero-hepatic circulation rather
than preventing hemolysis.
In a study of adult patients with Gilbert syndrome,
oral zinc sulfate at a dose of 100 mg daily for 7 days resulted in a
significant decline in serum bilirubin levels [8]. Our results are at
variance with this study. It may be speculated that zinc is ineffective
in disrupting entero-hepatic circulation of bilirubin in the face of
high bilirubin loads which occur in newborns. There is also a
possibility that zinc may interact with intestinal flora to modulate
entero-hepatic circulation of bilirubin. The relative paucity of
bacterial flora in newborns might have contributed to the lack of effect
of zinc in the present study. Further, neonatal hyperbilirubinemia is
typically a multifactorial disorder with different mechanisms
contributing to hyperbilirubinemia risk, and thus zinc may have a
limited impact in this age group.
Though the duration of phototherapy was less by 21.3
h in the zinc group, the difference was not statistically significant.
This could be due to small sample size as only 21 newborns in zinc group
and 18 newborns in placebo group required phototherapy. This difference
may be clinically relevant and further studies with a larger sample are
warranted to clarify this issue. However, the present study was limited
by the fact that we estimated TSB levels at fixed time intervals and
this might have influenced the duration of phototherapy in present
study. Future study design should include transcutaneous bilirubin
estimation at more frequent intervals to detect the effect of zinc
supplementation on the duration of phototherapy.
To conclude, oral zinc sulfate is not beneficial in
the treatment of moderate grade idiopathic neonatal hyperbilirubinemia
in term and near-term newborns.
Contributors: AK: study planning, design and
writing of the manuscript; NKB: data collection, analysis and writing of
the manuscript; SB: study planning and writing of the manuscript. RKA:
data analysis and writing of manuscript.
Funding: Zinc sulfate and placebo were procured
from Apex Pharmaceuticals Private Limited, Chennai, India.
Competing interest: None stated.
|
What is Already Known?
• Zinc salts can sequester unconjugated
bilirubin in vitro and zinc sulfate can inhibit biliary
secretion of bilirubin in experimental animals by reducing its
enterohepatic circulation.
What This Study Adds?
• Oral zinc sulfate (10 mg/day) is not
effective in the management of idiopathic neonatal
unconjugated hyperbilirubinemia.
|
References
1. Xiong T, Qu Y, Cambier S, Mu D. The side effects
of phototherapy for neonatal jaundice: what do we know? What should we
do? Eur J Pediatr. 2011;170:1247-55.
2. Smits-Wintjens VE, Rath ME, van Zwet EW, Oepkes D,
Brand A, Walther FJ, et al. Neonatal morbidity after exchange
transfusion for red cell alloimmune hemolytic disease. Neonatology.
2013;103:141-7.
3. Vitek L, Zelenka J, Zadinova M, Malina J. The
impact of intestinal microflora on serum bilirubin levels. J Hepatol.
2005;42:2338-43.
4. Vitek L, Muchova L, Zelenka J, Zadinova M, Malina
J. The effect of zinc salts on serum bilirubin levels in
hyperbilirubinemic rats. J Pediatr Gastroenterol Nutr. 2005;40:135-40.
5. Rana N, Mishra S, Bhatnagar S, Paul V, Deorari AK,
Agarwal R. Efficacy of zinc in reducing hyperbilirubinemia among at-risk
neonates: a randomized, double-blind, placebo-controlled trial. Indian J
Pediatr. 2011;78:1073-8.
6. Bhutani VK, Johnson L, Sivieri EM. Predictive
ability of a predischarge hour-specific serum bilirubin for subsequent
significant hyperbilirubinemia in healthy term and near-term newborns.
Pediatrics. 1999;103:6-14.
7. American Academy of Pediatrics Subcommittee on
Hyperbilirubinemia. Management of hyperbilirubinemia in the newborn
infant 35 or more weeks of gestation. Pediatrics. 2004;114:297-316.
8. Mendez-Sanchez N, Roldan-Valadez E, Gonzalez V, Roldan-Valadez E,
Flores MA, Uribe M. Zinc sulfate inhibits the enterohepatic cycling of
unconjugated bilirubin in subjects with Gilbert’s syndrome. Ann Hepatol.
2002;1:40-43.
|
|
|
 |
|

