|
Cellular and humoral immune
abnormalities with T-cell dysfunction and imbalance of Th1/Th2 cytokines
are proposed to play an important role in the development of HSP [1].
The T-cell immunoglobulin- and mucin-domain-containing molecules-1
(Tim-1) is expressed on T helper type 2 (Th2) and plays an important
role in the activation of T cells and Th2-mediated immune responses [2].
Recently, Wang, et al. [3] showed the expression of Tim-1 and
relevant cytokines in peripheral blood mononuclear cells in patients
with lupus erythematosus, suggesting that the molecule may play an
important role in the pathogenesis of autoimmune diseases.
HSP has been proved to be initiated and mediated by
autoreactive T cells triggered by uncertain etiology. However, it has
not been determined whether the Tim gene-family plays a role in the
development of HSP. In the present study, we examined the expression of
Tim-1 and relevant cytokines in peripheral PBMC in HSP and healthy
children to investigate the role of Tim-1 and its modulating the balance
of Th1/Th2 cells in the disease.
Methods
Twenty Chinese children (mean age 8.75 ± 2.20 years
old, range 6-13 years old) with acute onset and/or active presentation
of HSP at this hospital during January 2007 to June 2009 were included.
The diagnosis of HSP was based on standard classification criteria [4].
Fifteen healthy subjects (mean age 9.35 ± 2.30 years,
range 6-13 years old) were recruited as normal controls. Informed
consent and institutional approval were obtained for the study. The
activity of HSP was scored [5]. The mean clinical score was 4.50 ± 1.15.
PBMC were isolated from peripheral blood following
standard protocols. PBMC were harvested and the proportion of viable
cells assessed by trypan blue exclusion. More than 95% of the cells were
viable. Real time quantitative polymerase chain reaction (PCR) was
performed to determine on RNA expression for Tim-1.Total RNA were
isolated from PBMCs using Trizol reagent (Invitrogen, Shanghai, China).
Total RNA (1 µg) was reverse transcribed into cDNA using AMV reverse
transcriptase (Fermentas,). Primers for Tim-1 and -actin were as
follows:Tim-1, Forward: 5’-CCAGTAG CCACTTC ACCATCTT-3’; Reverse:
5’-TGTTATTC CAAA GGCC ATCTGA-3’(160bp); ²-actin, Forward:
5’-TGACGTGGACATCCGCAAAG-3’; Reverse: 5’-CTGGAAGGTGGACAGCGAGG -
3’(205bp). Conditions for the PCR were as follows: 95ºC for 4 minutes,
followed by 35 cycles for Tim-1 or 30 cycles for
b-actin. The PCR
products were run on an agarose gel and were in all cases confined to a
single band of the expected size (data not shown). 2–DCT
was used to figure the expression value of Tim-1. Blood levels of tumor
microsibfactor a
(TNF-a), IL-4
and IgA1 were estimated by ELISA [6].
Differences in relative mRNA levels of Tim-1 and
cytokines were tested for significance using Mann–Whitney test.
Correlations between Tim-1 and cytokine levels were analyzed with
Spearman’s rank test; P value<0.05 was considered significant.
Results
The expression of Tim-1 in HSP patients was
significantly higher than the controls (2.05 ± 0.83 vs 0.67 ±
0.09) (Table I). There was a significant positive
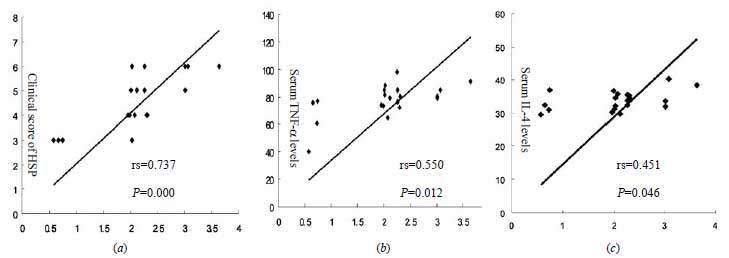
correlation between Tim-1 expression and active HSP (Fig. 1a).
In addition, there was a significant increase in expression of TNF-a
and IL-4 in HSP patients compared with the controls (33.66 ± 2.96 vs
5.19 ± 2.35 pg/mL; and 77.42 ± 12.21 vs 24.29 ± 4.37 ng/mL,
respectively). The levels of TNF-a
and IL-4 correlated with Tim-1 expression in HSP
patients (Fig. 1b and c). The serum
IgA1 levels were elevated in HSP patients with compared with healthy
controls (1.48 ± 0.40 vs 0.43 ± 0.13 mg/mL) (P <0.01) and
there was significant correlation between the serum IgA1 and Tim-1mRNA
in HSP patients.
TABLE I Tim-1 Expression and Serum TNF-a, IL-4 and IgA1 Levels in the Two Groups
|
Group |
n |
IgA1 (mg/mL) |
TNF-a (pg/mL) |
IL-4 (ng/mL) |
Tim-1 expression* |
| Controls |
15 |
0.43 ± 0.13 |
5.19 ± 2.35 |
24.29 ± 4.37 |
0.67 ± 0.09 |
| Henoch-Schonlein
Purpura |
20 |
1.48 ± 0.40 |
33.66 ± 2.96 |
77.42 ± 12.21 |
2.05 ± 0.83** |
|
*Measured in peripheral blood mononuclear
cells using quantitative real-time reverse
transcription-polymerase chain reaction; Data are expressed as
mean ± SD; P<0.01 compared with the controls for all
measurements; Tim-1; T-cell immunoglobulin – and mucin-domain –
containing molecule -1; TNF- a
: Tumour necrosis factor –
a; IL-4:
Interlactin – 4; IgA 1: Immunoglobulin A1.
|
 |
|
Fig. 1 Correlation between Tim-1
expression and (a) clinical score of Henoch-Schonlein purpura
(b) Interleukin-4, and (c) Tumour necrosis factor
a .
|
Discussion
Tim molecules, constitute a family of molecules
expressed on T cells, and are associated with the regulation of Th2
immune responses [2]. In the present study, we found that Tim-1
expression was upregulated on PBMC from patients with HSP, correlated
with the clinical score.
Two studies recently demonstrated that Tim-1
engagement can promote allograft acceptance or rejection, effects that
are dependent on regulatory T cells [7]. In vitro T cell
stimulation with an agonistic anti-Tim-1 Ab increased the number of
interleukin (IL-17)- and interferon (IFN- a)-producing
cells [8]. Mice treated with an anti–Tim-1 Ab or a Tim-1 extracellular
domain protein showed attenuated development of antigen-induced airway
inflammation and of contact or delayed-type hyper-sensitivity responses
[9]. The immuno-inflammatory response induced by IgA1-containing immune
complexes is considered important in the pathogenesis of HSP [1]. We
also found the serum IgA1 levels were significantly increased in HSP
patients. Moreover the high IgA1 was closely related with Tim-1
expression, indicating that upregulation in Tim-1 expression may a cause
of triggering the activation of B cells and IgA1 secretion.
There was a significant increase in the expression of
serum TNF- a
and IL-4 in HSP patients. The expression TNF-a
of and IL-4 correlated well with Tim-1. Taken
together, our results indicate an increased Th2 response in HSP patients
associated with a upregulation of Tim-1 expression. Our results suggest
that Tim-1 may act as a potent regulator of Th2 cells by regulating
their cytokines.
Contributors: All the authors were
involved in all aspects of the study. LPY shall be the guarantor for the
study.
Funding: This project was supported by the
scientific and technological key project of Anhui Province, No
07010302198.
Competing interests: None stated.
|
What This Study Adds?
• High Tim-1 expression was correlated with
blood levels of TNF-α,
IL-4 and IgA1 in patients with HSP, which may be involved in the
pathogenesis of HSP.
|
References
1. Xie HT, Zhang JB. Progress in research on
immunobiology of Henoch-Schonlein purpura. Journal of Inner Mongolia
University for Nationalities. 2009;24:453-6.
2. Kane LP. TIM family proteins and autoimmunity.
Autoimmunity. 2007;40:405-8.
3. Wang Y, Meng J, Wang X, Liu S, Shu Q, Gao L, et
al. Expression of human TIM-1 and TIM-3 on lymphocytes from systemic
lupus erythematosus patients. Scand J Immunol. 2008; 67:63-70.
4. Ozen S, Ruperto N, Dillon MJ, Bagga A, Barron K,
Davin JC, et al. EULAR/PReS endorsed consensus criteria for the
classification of childhood vasculitides. Ann Rheum Dis. 2006;65:936-41.
5. Foster BJ, Bernard C, Drummond KN, Sharma AK.
Effective therapy for severe Henoch-Schonlein purpura nephritis with
prednisone and azathioprine: a clinical and histopathologic study. J
Pediatr. 2000;136:370-5.
6. Zhang QÿLu LÿLin D. Levels and significance of
IgA1 in serum and skin lesions of pediatric patients with anaphylactoid
purpura. Chinese J Dermatol.2008;41:55-6.
7. Umetsu SE, Lee WL, McIntire JJ, Downey L,
Sanjanwala B, Akbari O, et al. TIM-1 induces T cell
activation and inhibits the development of peripheral tolerance. Nat
Immunol. 2005;6:447-54.
8. Fukushima A, Sumi T, Fukuda K, Kumagai N, Nishida
T, Akiba H, et al. Antibodies to T-cell Ig and mucin
domain-containing proteins (Tim)-1 and -3 suppress the induction and
progression of murine allergic conjunctivitis. Biochem Biophy Res Commun.
2007;353:211-6.
9. Ueno T, Habicht A, Clarkson MR, Albin MJ, Yamaura
K, Boenisch O, et al. The emerging role of T cell Ig mucin 1 in
alloimmune responses in an experimental mouse transplant model. J Clin
Invest. 2008;118:742-51.
|

