|
|
|
Indian Pediatr 2012;49:
449-454 |
 |
Vitamin D Supplementation for Severe Pneumonia
– A Randomized Controlled Trial
|
|
Nidhi Choudhary and Piyush Gupta
From Department of Pediatrics, University College of
Medical Sciences and Guru Teg Bahadur Hospital,
Dilshad Garden, Delhi, India.
Correspondence to: Dr Piyush Gupta, Block R-6A,
Dilshad Garden, Delhi 110 095, India.
Email:
[email protected]
Received: March 23, 2011;
Initial review: April 13, 2011;
Accepted: July 09, 2011.
Published online: 2011 August 15.
S097475591100214-1
|
Objective: To determine the role of oral vitamin D supplementation
for resolution of severe pneumonia in under-five children.
Design: Randomized, double blind,
placebo-controlled trial.
Setting: Inpatients from a tertiary care
hospital.
Participants: Two hundred children [mean (SD)
age: 13.9 (11.7) months; boys: 120] between 2 months to 5 years with
severe pneumonia. Pneumonia was diagnosed in the presence of fever,
cough, tachypnea (as per WHO cut-offs) and crepitations. Children with
pneumonia and chest indrawing or at least one of the danger sign
(inability to feed, lethargy, cyanosis) were diagnosed as having severe
pneumonia. The two groups were comparable for baseline characteristics
including age, anthropometry, socio-demographic profile, and clinical
and laboratory parameters.
Intervention: Oral vitamin D (1000 IU for <1 year
and 2000 IU for >1 year) (n=100) or placebo (lactose) (n=100)
once a day for 5 days, from enrolment. Both the groups received
antibiotics as per the Indian Academy of Pediatrics guidelines, and
supportive care (oxygen, intravenous fluids and monitoring).
Outcome variables: Primary: time to
resolution of severe pneumonia. Secondary: duration of
hospitalization and time to resolution of tachypnea, chest retractions
and inability to feed.
Results: Median duration (SE, 95% CI) of
resolution of severe pneumonia was similar in the two groups [vitamin D:
72 (3.7, 64.7-79.3) hours; placebo: 64 (4.5, 55.2-72.8) hours]. Duration
of hospitalization and time to resolution of tachypnea, chest
retractions, and inability to feed were also comparable between the two
groups.
Conclusion: Short-term supplementation with oral
vitamin D (1000-2000 IU per day for 5 days) has no beneficial effect on
resolution of severe pneumonia in under-five children. Further studies
need to be conducted with higher dose of Vitamin D or longer duration of
supplementation to corroborate these findings.
Key words: ARI, India, Pneumonia, Treatment, Vitamin D.
|
|
Pneumonia is one of the leading causes of
childhood mortality worldwide; accounting for nearly one-fifth of the
under-five deaths in India [1]. Besides antibiotics, zinc and vitamin A
have been suggested to have beneficial role in children with severe
pneumonia [2-5]. Recent research indicates that Vitamin D may have a
potential role in protection from acute respiratory tract infections by
increasing the body’s production of naturally acting antibiotics [6].
Vitamin D has also been found to have immune modulating properties by
virtue of its ability to induce monocyte differentiation and inhibit
lymphocyte proliferation [6-10]. Vitamin D has antimicrobial properties
as it stimulates phagocytosis-dependent and antibody-dependent
macrophages [6].
1,25-dihydroxy vitamin D3
also acts upon T and B cells and can modulate functions of lymphocytes
that produce cytokines and antibodies [8]. Severe deficiency of vitamin
D leads to chest wall deformity and hypotonia, leading to reduced lung
volume, poor compliance of the chest wall, atelectasis and fibrosis
[11]. An Ethiopian study has found a 13-fold higher incidence of severe
vitamin D deficiency in children with pneumonia, as evident clinically
by presence of rickets [12].
Despite evidence for a positive correlation between
vitamin D deficiency and incidence of pneumonia [11], data regarding the
direct effect of vitamin D supple-mentation in acute attacks of
pneumonia is limited to only one study [13]. We conducted this trial to
test our hypothesis that vitamin D supplementation decreases the
duration of resolution of severe pneumonia in children under five years
of age.
Methods
A double blind, randomized, placebo-controlled trial
was conducted in children between 2 months and 5 years admitted with a
diagnosis of severe pneumonia in a tertiary care hospital. Permission
was obtained from the institutional ethical committee and written
informed consent obtained from the caregiver of the participant.
Enrolment: The sample size was based on a
hospital based randomized control trial on the role of zinc for severe
pneumonia in very young children [3]. A total of 98 patients were
required in each group to detect a meaningful difference of one day in
resolution of severe pneumonia with standard deviation of 2.5, type 1
a error 0.05
and power of 80%. Eligible subjects included all children between 2
months – 5 years with a clinical diagnosis of severe pneumonia,
presenting to pediatric emergency department. Children with fever,
cough, tachypnea and crepitations were diagnosed with pneumonia.
Tachypnea was defined as respiratory rate
³50/min in children
between 2-12 months and ³40/min
in 1-5 years age group [14]. Those with pneumonia and either chest
indrawing or at least one other danger sign (inability to feed,
lethargy, and cyanosis) were diagnosed as having severe pneumonia.
Children with severe wasting (weight for height <3SD), chronic
illnesses, previous history of vitamin D intake over last 4 weeks, and
known asthmatics were excluded.
Randomization and blinding: Randomization was
done according to computer generated random number table. Allocation
concealment was done by sealed envelope technique. Both the caretaker
and subject were blind regarding the content of the drug been given.
Each sachet of vitamin D contained 1000 IU of drug whereas the placebo
sachet contained lactose. One sachet of vitamin D (60,000 IU) weighing
1g was mixed with 11 gm of lactose, total weight being 12g. Of this, 60
doses were taken each weighing 200 mg so that each dose carried 1000 IU
of vitamin D. Both looked alike in terms of appearance, taste and color.
The code key was opened only after the intervention, data collection,
follow up and tabulation were completed.
Initial data collection: A detailed history
(including socio-demographic profile, feeding history, immunization,
past history of pneumonia, fuel use or smoking) was taken and clinical
examination (including anthropometry as per tools and standard
techniques) [15] was
performed for each of the participant. Respiratory rate were counted for
full 60 seconds, after removing all clothes from the torso and also
chest indrawing was observed at the same time. Average of two readings
was recorded. If the readings differed by more than 5 breaths per
minute, a third reading was taken and the two closest readings averaged.
Child had to be awake and not crying during these measurements. Axillary
temperature was taken with a standardized mercury thermometer. Fever was
defined as temperature >38ºC. Baseline oxygen saturation was measured in
room air using a pulse oximeter with a probe on a finger or toe. Hypoxia
was defined as oxygen saturation <95% in room air [3]. Weight and height
were measured using standard techniques. Venous blood sample was
obtained for hemogram, blood sugar, electrolytes, C-reactive proteins
(latex slide test, >0.8mg/dL taken as positive), calcium, phosphate and
alkaline phosphatase. All data including respiratory rate, retractions,
oxygen saturation, fever, feeding, cyanosis and mental status were
recorded.
Intervention: Children were randomized to receive
either vitamin D or placebo as a single oral dose every day, for five
days from enrollment. Authors were not involved with the randomization
process. Vitamin D was given as 1000 IU to children less than one year
old and 2000 IU children between 1-5 years of age. The drug/placebo was
dispensed in milk and administered orally within four hours of
admission. This was followed by once-a-day dosing for next 4 days. Those
unable to take orally were given the drug by nasogastric tube. The drug
was repeated if an episode of vomiting occurred within 15 minutes of
ingestion of drug. Both the groups received antibiotics as per Indian
Academy of Pediatrics (IAP) guidelines [16] and supportive care (oxygen,
intravenous fluids and monitoring). Children with associated wheezing
received salbutamol nebulization twice at an interval of 20 minutes.
Clinical monitoring: Data including respiratory
rate, retractions, oxygen saturation, fever, feeding, cyanosis and
mental status were recorded every 8 hourly. Worsening of any one sign
was qualified as ‘deteriorating’ condition and no change in any sign as
failure to improve. Child was reclassified from severe pneumonia to
pneumonia (non severe) when chest indrawing and hypoxia (saturation less
than 95% on room air) were absent for 24 consecutive hours and
respiratory rate was less as per age cut off, at which oral antibiotic
was started. Oral feeding was also started at this point of time. If any
signs recurred, the child was reclassified as severe until these
conditions were met. All children received a minimum of 5 days of
IV/oral antibiotics. Children were discharged when tachypnea had
subsided for a minimum of 24 hours. Subjects with clinical rickets were
given a mega dose of vitamin D (6,00,000 IU) at the time of discharge.
All children were observed for adverse effects and compliance.
Primary outcome variable: The primary outcome of
interest was the time to resolution of severe pneumonia. Resolution of
severe pneumonia was considered when lower chest retraction and the
danger signs (inability to feed, lethargy, cyanosis or hypoxia) were no
longer present.
Secondary outcome variables: The secondary
outcome variables included the duration of hospitalization, and time to
resolution of tachypnea (respiratory rate cut off for severe pneumonia
as per age), chest retractions, and inability to feed. The duration of
hospitalization was defined as the time (in hrs) between study enrolment
and discharge. The patient was considered fit for discharge when he/she
was afebrile (axillary temperature <37.5ºC), tachypnea had subsided,
there were no chest indrawings, and oral feeding had resumed, for a
minimum period of 24 hours.
Statistical analysis: Baseline characteristics
between the two groups were compared using chi-square/Fisher’s test for
categorical variables and unpaired t test for quantitative
variables, as these were normally distributed. The effect of vitamin D
supplementation on outcome variables was analyzed on intention to treat
basis. Kaplan Meier survival function plots were constructed to compare
median duration of each outcome variable between the two groups using
the log rank test. P <0.05 was taken as significant. The data
were analyzed by using SPSS software.
Results
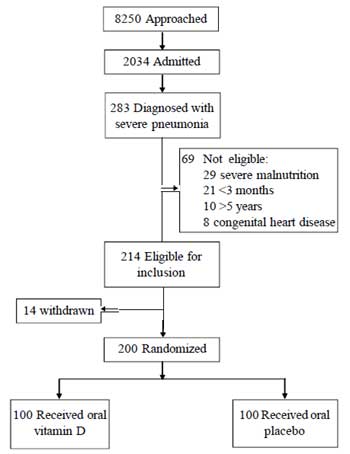
Overall, 283 children were diagnosed with severe
pneumonia during the study period, of which, 200 were randomized (Fig.
1). The median (IQR) age of study subjects was 10 (5-18) months.
Number of children below 2 years was comparable in the two groups
(vitamin D: 84; placebo: 85). The two groups were also comparable for
other baseline characteristics including socio demographic profile,
anthropometry, and clinical and laboratory parameters (Table I
and II). Only 5 children had clinical evidence of rickets; 2
in vitamin D and 3 in placebo group. Fever, cough, coryza, irritability
and decreased oral acceptance were seen in all patients admitted and
enrolled for the study. The median (IQR) duration of illness at
presentation was 5 (4-7) days. Wheeze was present in almost 1/3 rd
of the subjects (33.5%); 32 children in vitamin D group and 35 in
placebo group. Cyanosis was not present in any case on presentation. The
mean oxygen saturation was 92.8% ± 2.4. C-reactive protein (CRP) was
positive in 161/197 cases (81.7%). Of these, 82 cases were in vitamin D
group and 79 cases in the placebo group.
TABLE I Baseline Characteristics of The Study Population
| Parameter |
Vitamin D |
Placebo |
|
n (%) |
n (%) |
| Socioeconomic status |
| Upper |
0 |
0 |
| Upper middle |
10 (10%) |
4 (4%) |
| Lower middle |
22 (22%) |
23 (23%) |
| Upper lower |
66 (66%) |
72 (72%) |
| Lower |
2 (2%) |
1 (1%) |
| Residence |
| Urban |
24 (24%) |
19 (19%) |
| Rural |
29 (29%) |
34 (34%) |
| Slum |
47 (47%) |
47 (47%) |
| Passive smoking |
28 (28%) |
30 (30%) |
| Use of wood /coal / kerosene |
28 (28%) |
32 (32%) |
| Immunization |
| Complete |
51 (51%) |
38 (38%) |
| Partial |
34 (34%) |
37 (3%) |
| Unimmunized |
15 (15%) |
25 (25%) |
| Past history of pneumonia |
30 (30%) |
33 (33%) |
| History of TB contact |
1 (1%) |
0 |
TABLE II Baseline Characteristics, Anthropometry, Vital Signs And Laboratory Parameters
in the Vitamin D versus Placebo Group
Parameters
|
Vitamin D
(Mean±SD) |
Placebo
(Mean±SD) |
|
Age (mo) |
14.1±12.2 |
13.8±11.4 |
|
Weight (kg) |
7.5±2.5 |
7.3±2.7 |
|
Length (cm) |
70.0±10.6 |
70.1±11.9 |
|
Head circumference (cm) |
44.6±3.2 |
44.3±3.7 |
|
Chest circumference (cm) |
44.6±4.1 |
44.4±4.2 |
|
Mid-arm circumference (cm) |
11.2±1.4 |
11.1±1.4 |
|
Breastfeeding* (mo) |
7.7±5.8 |
8.5±5.9 |
|
Exclusive breastfeeding* (mo) |
3.9±1.7 |
3.9±2.4 |
|
Complementary feeding# (mo) |
7.8±2.0 |
8.7±2.3 |
|
Respiratory rate (breaths/min) |
69.2±9.1 |
68.2±8.9 |
|
SpO2 |
92.8±2.6 |
92.9±2.3 |
|
Hemoglobin (g/dL)
|
9.3±1.7 |
9.0±1.5 |
|
TLC (/cumm) |
11938.6±5320 |
12910±6640 |
|
S. Calcium (mg/dL) |
9.2±0.81 |
9.2±0.75 |
|
S. Phosphorus (mg/dL) |
5.1±1.4 |
5.3±1.8 |
|
S. ALP (IU/L) |
292.6±136.7 |
356.1±244.7 |
|
* Duration; #Age at initiation of |
 |
|
Fig. 1 Flow chart depicting the
inclusion of the study subjects.
|
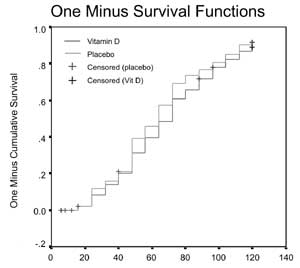
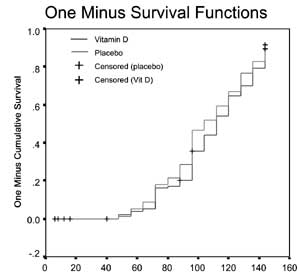
The median duration for resolution of severe
pneumonia and hospitalization was comparable in the two groups (Fig
2a, 2b). The two groups were also comparable for
time taken for resolution of tachypnea, chest retractions, hypoxia,
fever, inability to feed and lethargy/irritability (Table III).
 |
 |
|
Fig. 2 (a) Time to resolution of
severe pneumonia (hours); (b) Duration of
hospitalization (hours).
|
TABLE III Resolution of Symptoms (Hours) in Vitamin D and Placebo Group
|
|
Vitamin D |
Placebo |
*P value |
|
n |
Median (IQR) |
n |
Median (IQR) |
|
|
Resolution of
|
|
|
|
|
|
|
Severe pneumonia (h) |
87 |
72 (48-96) |
86 |
64 (48-88) |
0.33 |
|
Tachypnea (h) |
87 |
72 (56-104) |
84 |
72 (48-98) |
0.33 |
|
Chest retractions (h) |
87 |
64 (40-88) |
86 |
64 (40-88) |
0.38 |
|
Hypoxia (h) |
87 |
16 (8-24) |
86 |
16 (8-24) |
0.86 |
|
Fever (h) |
87 |
80 (64-104) |
85 |
72 (56-104) |
0.52 |
|
Inability to feed/lethargy (h) |
87 |
64 (48-88) |
86 |
56 (48-72) |
0.21 |
|
Duration of hospitalization (h) |
87 |
112 (96-136) |
86 |
104 (88-128) |
0.29 |
|
*Group comparison done with survival analysis using log-rank
test. |
A total of 140 children (vitamin D 68, placebo 72)
received first line antibiotics, whereas in 60 children, second line
antibiotics had to be started. Of those who received second line
antibiotics, 32 children were in vitamin D group and 28 in placebo
group. Staphylococcal coverage was given in 25 children, of which 18
were in vitamin D group and 7 in placebo group. Of 200 children, 97
children in vitamin D group and 94 children in placebo group were
finally discharged. Of the 191 children discharged, 28 children (11
vitamin D, 17 placebo) were discharged within 72 hrs, 95 (47 vitamin D,
48 placebo) were discharged between 72-120 hrs while 65 children (34
vitamin D, 31 placebo) were discharged after 120 hours of
hospitalization.
Overall 173 (86.5%) children improved (vitamin D: 87;
placebo: 86) and 23 (11.5%) remained in the same condition. Worsening
occurred in 4 (2%) children only. Two children died, 1 each in vitamin D
and placebo group. A total of 7 children could not complete the study as
parents left against medical advice (Fig. 1). There was no
difference between the two groups in the proportion of children who
improved.
A total of 191 children received all five doses of
the drug. No major adverse effects were noted. Only one child had a
single episode of vomiting and another complained of diarrhea that
lasted two days. Both the children belonged to the vitamin D group.
Discussion
The results indicate that short-term supplementation
with vitamin D does not decrease the duration of resolution of severe
pneumonia, duration of hospitalization, and time taken for resolution of
individual symptoms of severity of pneumonia in under-five children.
Vitamin D supplementation was well tolerated in all age groups without
showing any major side effects. These results are important; however,
they need further deliberations. Our study has certain limitations and
thus it may not be justified to generalize the results to all acute
lower respiratory tract infection at all ages and all settings.
Due to safety concerns, we kept the doses in our
study lower than the No Observed Adverse Effect Level (NOAEL; 2400IU)
and Lowest Observed Adverse Effect Level (LOAEL; 3800 IU) [17]. A higher
dose was not administered because of lack of safety data in children and
absence of vitamin D level monitoring. It is possible that a larger dose
is required to demonstrate the therapeutic effects of vitamin D in
infection. Initially it was thought that people living in areas with
poor sun exposure are affected but now there is evidence that vitamin D
deficiency is rampant even in tropical countries. Mothers and their
breast fed infants have also been found to be severely deficient in
vitamin D [18]. Thus it may be possible that the recruited children were
already deficient in vitamin D and our doses were too less to cause an
impact. This could have been avoided by measuring vitamin D levels but
due to financial constraints, the levels could not be checked and hence,
we could not categorize the therapeutic effect separately for vitamin D
replete and deplete subjects. Previous studies have also shown that
after receiving single large doses, some of the cases took nearly 2
months for the peak concentration to be achieved [19]. Thus it could be
possible that with the daily doses given in our study, the blood levels
of vitamin D might still be sub-optimal for the effects to occur.
We could not find any trial that evaluated the
therapeutic role of daily supplementation of vitamin D in severe
pneumonia. Only a single trial till now has ascertained the role of
vitamin D in pneumonia which studied 453 children, of which 224 children
received single dose of 100,000 IU of oral vitamin D and 229 children
received placebo. No significant difference was noted in the duration of
recovery from severe pneumonia, but recurrence was lower in vitamin D
supplemented group within 90 days of supplementation [13].
We conclude that short-term supplementation with 2000
IU of vitamin D given orally for five days in severe pneumonia to
under-five children has no beneficial effect on resolution of severe
pneumonia. However, further studies needs to be conducted with higher
doses of vitamin D and longer duration of supplementation and follow-up,
to ascertain the therapeutic as well as prophylactic role of vitamin D
for childhood pneumonia. Vitamin D estimation also needs to be done and
the response segregated for vitamin D deplete and replete children.
Contributors credit: NC: Collected the data and
wrote the initial manuscript; PG: designed the study, searched the
literature and critically analyzed the data and manuscript. Both authors
approved the final manuscript.
Funding: None; Competing interests: None
stated.
|
What is Already Known?
• Vitamin D deficiency predisposes to
infections; vitamin D acts as immune modulator.
What This Study Adds?
• Vitamin D supplementation at doses of 1000 IU and 2000 IU
for children <1 year and > year, respectively given daily for 5
days has no beneficial effect in severe pneumonia.
|
References
1. UNICEF/WHO. Pneumonia: The Forgotten Killer of
Children. Geneva: WHO; 2006.
2. Zinc Investigators’ Collaborative Group.
Prevention of diarrhea and pneumonia by zinc supplementation in children
in developing countries: pooled analysis of randomized controlled
trials. J Pediatr. 1999;135:687-97.
3. Brooks WA, Yunus M, Santoshan M, Wahed MA, Nahar
K, Yeasmin S, et al. Zinc for severe pneumonia in very young
children; double- blinded placebo- controlled trial. Lancet.
2004;363:1683-88.
4. Aggarwal R, Sentz J, Miller MA. Zinc
administration in prevention of childhood diarrhea and respiratory
illness: A meta-analysis. Pediatrics. 2007;6:1120-30.
5. Vitamin A and Pneumonia Working Group. Potential
interventions for the prevention of childhood pneumonia in developing
countries: a meta-analysis of data from field trials to assess the
impact of vitamin A supplementation on pneumonia morbidity and
mortality. Bull WHO. 1995;73:609-19.
6. Rallof J. The antibiotic vitamin. Deficiency in
vitamin D may predispose people to infection. Science. 2006;170;312.
7. Liu PT, Stenger S, Li H, Wenzel L, Tan BH ,
Krutzik SR, et al. Toll-like receptor triggering of a
vitamin D-mediated human antimicrobial response. Science.
2006;311:1770-3.
8. Lemire JM, Adams JS, Kermani- Arab V, Bakke AC,
Sakai R, Jordan SC. 1, 25-dihydroxyvitamin D3 suppresses human T helper
inducer lymphocyte activity in vitro. J Immunol. 1985;134:3032-5.
9. Wang TT, Nestel FP, Bourdeau V, Nagai Y, Wang Q,
Liao J, et al. Cutting edge: 1, 25-dihydroxyvitamin D3 is a
direct inducer of antimicrobial peptide gene expression. J Immunol.
2004;173:2909-12.
10. Gombart AF, Borregaard N, Koeffler HP. Human
cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the
vitamin D receptor and is strongly upregulated in myeloid cells by
1,25-dihydroxyvitamin D3. FASEB. 2005;19: 1067-77.
11. Wayse V, Yousafzai A, Mogale K, Filteau S.
Association of subclinical vitamin D deficiency with severe acute lower
respiratory infection in Indian children under 5 years. Eur J Clin Nutr.
2004;58:563-7.
12. Auss-Kettis A, Bjornesjo KB, Manneimer E, Cvibach
T, Clark P, Mammo D. The occurrence and clinical picture of
disease in a clinic in Addis Ababa. Ethiopia Med J. 1965;3:109-21.
13. Manaseki-Holland S, Masher I, Bruce J, Mughal Z,
Chandramohan D, Walraven G. Effects of vitamin D supplementation to
children diagnosed with pneumonia in Kabul: a randomized controlled
trial. Trop Med Int Health. 2010;15:1148-55.
14. WHO Child and Adolescent Health and Development
(CAH). Integrated management of neonatal and childhood illness and HIV.
Physician Chart Booklet. Ministry of Health & Family Welfare, Govt. of
India, 2007.
15. Physical status: The Use and Interpretation of
Anthro-pometry. Report of a WHO Expert Committee. Geneva: WHO; WHO
Technical Report Series No. 854. 1995.
16. Agarwal R, Singh V, Yewale V. RTI Facts. IAP
Consensus Guidelines on Rational Management of Respiratory Tract
Infections in Children. Mumbai: Indian Academy of Pediatrics; 2006.
17. Office of Dietary Supplements: NIH Clinical
Center. Dietary Supplement Fact Sheet: Vitamin D. Bethesda: National
Institutes of Health; 2007.
18. Saadi HF, Dawodu A, Afandi B, Zayed R, Benefict
S, Nagelkerke N, et al. Effect of combined maternal and infant
vitamin D supplementation on vitamin D status of exclusively breastfed
infants. Matern Child Nutr. 2009;5:525-32.
19. Veith R. Vitamin D supplementation, 25-hydroxyvitamin D
concentrations, and safety. Am J Nutr. 1999;69:842-56.
|
|
|
 |
|

