ongenital Hypothyroidism is the most common
preventable cause of mental retardation with an incidence of 1:2500 to
1:2800 live births in India [1, 2]. Clinical diagnosis is difficult at
birth and the time of initiation of therapy is a critical determinant of
outcome. In view of paramount importance of early diagnosis and
treatment, various screening programs were initiated [3,4]. In India,
ICMR introduced congenital hypothyroidism screening program in neonates
at various centers in 2007 [5,6]. Neonatal screening methods measure
Thyroid Stimulating Hormone level in either cord blood sample or that
obtained from heel prick sample at 3 to 4 days of life. When cord blood
Thyroid Stimulating Hormone (CB TSH) is measured for congenital
hypothyroidism screening, it has a high sensitivity but with a high
false positive rates [7].
Various maternal and perinatal factors are known to
affect the CB TSH levels [8]. There is a scarcity of Indian data on the
effects of various factors on CB TSH levels. This study presents an
analysis of various maternal and perinatal factors on CB TSH level.
Methods
This cross-sectional study was conducted in the
neonatology unit of department of pediatrics at Fortis Escorts hospital,
Faridabad, a tertiary-care hospital in Delhi NCR region. Priori
calculation of sample size to study 10 factors in multiple regression
model with a small (0.02) effect size and type 1 error of 5% (P<0.05)
and power of 80% yielded a minimum sample size of 818. The study was
planned to include consecutive 1000 live born neonates delivered at our
hospital from July 2009 onwards to account for a maximum 20% drop out/
consent withdrawal or sample processing issues. We planned to include
all live births in the hospital from July 2009 onwards. Exclusion
criteria were: neonates with major life threatening malformations; those
with antenatally detected central nervous system malformations; and
neonates whose mothers were on any antithyroid drugs.
An informed consent was obtained from either of the
parents. Antenatal and intra-partum information was noted from mother’s
medical record. Blood samples were drawn for blood group and TSH assay
as per unit’s protocol, through a 5 mL syringe from the maternal end of
the cord immediately after the cord was cut. The sample thus collected
was kept at room temperature of around 25°C and was transported to
laboratory within one hour. Neonates whose blood samples were not
processed for technical reasons were excluded from final analysis
(commonly due to inadequate amount hemolyzed sample). The sample was
analyzed within 3 hours using electrochemiluminescence immunoassay on
Cobas e 411 analyser with functional sensitivity of 0.014 microIU/mL All
neonates who had CB TSH values more than 20 were advised repeat TSH
assessment within 14 days of life.
The data were entered in Excel sheet and percentages
of various outcome measures were calculated using SPSS for Windows
version 12. The effect of various perinatal factors on the CB TSH levels
was analyzed using independent Kruskal Wallis and Mann Whitney tests to
define differences between groups and a P value of <0.05 was
defined as significant. The relationship between variables was first
analyzed using univariate analysis and all the variables were then taken
to multivariate regression along with demographic factors. The study was
approved by the hospital ethics committee.
Results
Of 1000 newborns enrolled, one was excluded on
clinical grounds (mother on anti thyroid dugs) while another 47 cord
blood samples could not be processed. Thus the study population
comprised of 952 subjects (Table I). The CB-TSH values
ranged between 1.01- 63.74 microIU/mL with median at 8.75 (IQR =
6.475-12.82). 109 out of 952 neonates (11.45%) had CB-TSH values >20
microIU/mL and 44 (4.6%) had values >30. CB TSH values were found to be
significantly raised in neonates delivered as first order compared to
multiparous mothers (higher order births) (P=0.005) and in babies
delivered by assisted vaginal delivery and normal delivery compared to
caesarean section (P<0.001). Also, neonates who had fetal
distress or non-progress of labour had significantly higher CB TSH than
those who were delivered by elective caesarean section; (P<0.001).
Requirement of resuscitation beyond the initial steps and low Apgar
scores of <7 at 1 minute also resulted in significantly raised CB TSH (P<0.001).
Male neonates had slightly increased CB TSH than their female
counterparts (P =0.031). It was noticed that maternal
hypothyroidism, maternal hypertension and neonates’ weight
appropriateness for gestation do not significantly affect the CB TSH
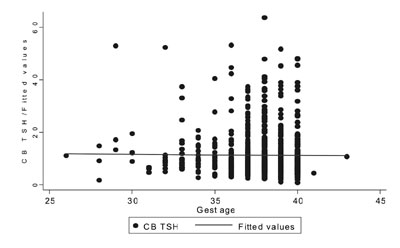
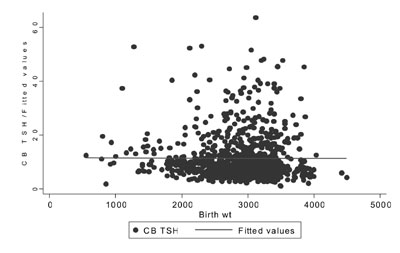
value (Table II). No correlation was found between CB TSH,
gestational age (r= -0.009) (Fig. 1) and birth weight (r=
-0.004) (Fig. 2). On multivariate analysis- requirement of
resuscitation, mode of delivery and fetal distress as indication for
lower segment cesarean section (LSCS) were found to be significant
factors.
TABLE I Profile of Subjects included in the Study
|
Characteristic
|
No (%) |
No (%) |
|
Birth Order |
|
First
|
544 |
57.1 |
|
Second |
342 |
35.9 |
|
Third or Higher |
66 |
6.9 |
|
Mode of Delivery
|
|
Normal Vaginal Delivery |
438 |
46.0 |
|
Assisted Vaginal Delivery |
60 |
6.3 |
|
Caesarean Section |
454 |
47.7 |
|
Indication of Caesarean Section |
|
Elective |
301 |
31.6 |
|
For Fetal Distress |
91 |
9.6 |
|
For Non Progress Of Labour |
53 |
5.6 |
|
Other |
9 |
0.9 |
|
Hypothyroid mother |
48 |
5.0 |
|
Euthyroid mother |
904 |
95.0 |
|
Maternal Pregnancy Induced Hypertension |
|
PIH |
67 |
7.0 |
|
Normotensive |
885 |
93.0 |
|
Weight Appropriateness for Age |
|
|
|
Small For Gestation |
31 |
3.3 |
|
Appropriate For Gestation |
873 |
91.7 |
|
Large For Gestation |
48 |
5.0 |
|
Male sex |
510 |
53.6 |
|
Resuscitation Required |
|
Routine Care |
802 |
84.2 |
|
Beyond Initial Steps |
150 |
15.8 |
|
APGAR Scores |
|
|
|
Less Than 5 |
28 |
2.9 |
|
5 or 6 |
84 |
8.8 |
|
7 or More |
840 |
88.2 |
|
Gestational Age |
|
Term (37-41 weeks) |
769 |
80.8 |
|
Preterm (<37 weeks) |
182 |
19.1 |
|
Preterm (<32 weeks) |
25 |
2.6 |
TABLE II Comparison of Cord Blood TSH Values in Accordance with Subject Characteristics
|
Characteristic |
Number
|
Median |
Interquartile |
P value
|
|
|
(microIU/mL) |
range |
|
|
First order birth
|
544 |
9.03 |
6.58 – 15.4 |
0.005 |
|
Higher order birth
|
408 |
7.47 |
4.83 – 10.56 |
|
|
Normal & assisted vaginal delivery
|
498 |
9.33 |
5.94 – 15.41 |
<0.001 |
|
Caesarean section
|
454 |
7.56 |
5.82 – 9.81 |
|
|
LSCS for fetal distress and non progress
|
153 |
7.69 |
5.3 – 11.05 |
<0.001 |
|
Elective LSCS
|
301 |
7.56 |
5.99 – 9.52 |
|
|
Hypothyroid mother |
48 |
8.76 |
6.47 -12.06 |
0.220 |
|
Euthyroid mother
|
904 |
8.05 |
5.47 – 11.72 |
|
|
PIH In mother
|
67 |
9.6 |
7.28 – 12.41 |
0.584
|
|
Normotensive mother
|
885 |
8.67 |
6.45 – 13.07 |
|
|
Male baby
|
510 |
9.26 |
6.63 – 13.35 |
0.031 |
|
Female baby
|
442 |
8.22 |
6.21 – 12.48 |
|
|
Small for gestation |
31 |
8.73 |
6.60 – 12.17 |
0.506 |
|
Appropriate for gestation |
873 |
8.8 |
6.51 – 12.87 |
|
|
Large for gestation
|
48 |
7.68 |
5.43 – 14.65 |
|
|
Resuscitation beyond initial steps
|
150 |
13.78 |
8.92 – 21.02 |
<0.001 |
|
Routine care
|
802 |
8.25 |
6.22 – 11.67 |
|
|
Apgar less than 7 |
112 |
12.42 |
8.18 – 19.23 |
<0.001 |
|
Apgar 7 or more
|
840 |
8.42 |
6.4 – 12.08 |
|
 |
|
Fig. 1 Correlation between gestational
age and CB TSH value.
|
 |
|
Fig. 2 Correlation between birth
weight and CB TSH value.
|
Discussion
Though the Screening for congenital hypothyroidism
will decrease the burden of mentally retarded children in the society,
the method of screening is not uniform [3]. Some countries use T4 while
others prefer TSH as the tool since maternal diseases affecting
placental dynamics influence T4 levels [9,10]. Few others use both T4
and TSH. Technically, using both T4 and TSH will be superior but would
increase the cost of screening. Most of the countries have accepted TSH
either through heel prick or through cord-blood as the screening method
for congenital hypothyroidism. Cord blood collection of sample is
preferred for its ease of collection of sample, lower rates of follow up
losses, more practical for mothers with short hospital stay following
delivery and its utility as an indicator of the prevalence of iodine
deficiency disorders [11,12].
Researchers have studied different CB TSH cut off
levels varying between 20-90 for recall with an objective to keep cost
of rescreening low and making it more cost effective. In Indian setup,
cord blood TSH value of >20µIU/mL is seen as safe cut off for recall
[13,14]. We, in the setting of tertiary care referral hospital, found
that 11.5% of all samples had values more than 20 µIU/mL which reflected
that a high recall rate is associated with CB TSH assessment. The only
other comparable study from a near similar geographical area [15] though
does not provide the numbers of patients with TSH levels more than 20
microU/mL, but reports the mean CB TSH as 10.6 +/- 6.7 microU/mL and
that their high risk patients (>6 % population) had a mean TSH above 20
microU/mL.
Changes in TSH levels in response to T3 and T4 blood
levels forms the basis of screening for congenital hypothyroidism
through CB TSH estimation. However, other factors may also influence TSH
levels. Various authors have correlated an increase in TSH values with
factors like birth asphyxia and difficult deliveries [15], perinatal
stress events [8], birth weight, male infant sex and instrumental
delivery [16], and negatively with cesarean sections as mode of delivery
[17]; but the mechanism are poorly understood.
The postnatal surge in TSH levels, common to all
newborns, is considered to be mediated through alpha adrenergic
stimulation following the cold stress [18]. In a study on neonatal rats,
it was demonstrated that perinatal hypoxia increases the secretion of
catecholamines [19]. Similarly, a surge in catecholamine secretion was
seen in human neonates during parturition; and this was more in
asphyxiated newborns and in vaginally delivered newborns compared to
those born by elective caesarean section [20]. Others too observed that
with perinatal hypoxia there is an increase in endogenous catecholamine
[21], which is more pronounced when the scalp PH is less than 7.26 [22].
This alpha adrenergic stimulation in turn might be responsible for the
observed increase in CB TSH in our subjects who had low Apgar scores,
required active resuscitation after birth, were born through vaginal
delivery or non-elective LSCS, and to primiparous mother. However, in
our study, no significant difference was found in CB TSH values in male
and female neonates; nor any positive correlation found with the birth
weight.
Unlike authors who observed a negative correlation of
serum TSH with gestational age [23], we did not find it to be
significant. At our center, we commonly give antenatal steroids before
premature deliveries and Dexamethasone has been shown to blunt the
release of catecholamine [24,25], which might have an effect on TSH
levels.
Perinatal stress factors and mode of delivery have a
significant impact on cord-blood TSH levels and any rise in cord blood
TSH should be seen in the light of these factors. The proportion would
be higher where high-risk pregnancies are delivered. Larger studies
should factor this impact and work out a correction in TSH cut off in
accordance to influencing factors if present. Many repeat evaluations of
thyroid function can thus be avoided, and would not only save the cost
but also would allay the anxiety of parents of neonates undergoing a
repeat/confirmatory test.
1. Desai MP, Colaco MP, Ajgaonkar AR, Mahadik CV, Vas
FE, Rege VV, et al. Neonatal screening for congenital
hypothyroidism in a developing country: problems and strategies. Indian
J Pediatr. 1987;54:571-81.
2. Desai MP, Upadhye P, Colaco MP, Mehre M, Naik SP,
Vaz FE, et al. Neonatal screening for congenital hypothyroidism
using the filter paper thyroxine technique. Indian J Med Res.
1994;100:36-42.
3. Newborn screening for congenital hypothyroidism:
recommended guidelines: American Academy of Pediatrics Section on
Endocrinology and Committee on Genetics and American Thyroid Association
Committee on Public Health. Pediatrics 1993;91:1203-9.
4. Nanayakkaral D, Wijekoon A, Jiffry N, Mudiyanse R,
Nilam J, Perera K, et al. Screening for congenital hypothyroidism
in government hospitals in Sri Lanka. Proceedings of the Peradeniya
University Research Sessions, Sri Lanka. 2007;12: 133-4.
5. Dutta R. ICMR to conduct first nationwide
newborn screening for genetic disorders. Express Health Care Management;
1st –15th September 2005.
6. Kapoor S, Kabra M. Newborn Screening: Current
Perspectives. Indian Pediatr. 2010;47:219-24.
7. Kaur G, Srivastav J, Jain S, Chawla D, Chavan
BS, Atwal R, et al. Preliminary report on neonatal screening for
congenital hypothyroidism, congenital adrenal hyperplasia and
glucose-6-phosphate dehydrogenase deficiency: A Chandigarh experience.
Indian J Pediatr. 2010;77:969–73.
8. Kim EY, Park SK, Song CH, Lim SC. Perinatal
factors affecting thyroid stimulating hormone (TSH) and thyroid hormone
levels in cord blood. Korean J Pediatr. 2005;48:143-7.
9. Franklin RC, Carpenter LM, O’Grady CM. Neonatal
thyroid function: influence of perinatal factors. Arch Dis Child.
1985;60:141-4.
10. Fuse Y, Wakae E, Nemoto Y, Uga N, Tanaka
M, Maeda M, et al. Influence of perinatal factors and sampling
methods on TSH and thyroid hormone levels in cord blood. Endocrinol
Japon. 1991;38:297-302.
11. Mu Li, Eastman CJ. Neonatal TSH Screening: is it
a sensitive and reliable tool for monitoring iodine status in
populations. Best Practice and Research Clinical Endocrinology and
Metabolism. 2010;24;63-75.
12. Kýslal F, Cetinkaya S, Dilmen U, Yasar H, Tezic
T. Cord blood thyroid stimulating hormone (TSH) and free T4 (fT4) levels
in Turkish neonates: Is iodine deficiency still a continuing
problem? Pediatrics International October 2010;5:762-8.
13. Manglik AK, Chatterjee N, Ghosh G. Umbilical cord
blood TSH levels in term neonates: A screening tool for congenital
hypothyroidism. Indian Pediatr. 2005;42:1029-32.
14. Ogunkeye OO, Roluga AI, Khan FA. Resetting
the Detection Level of Cord Blood Thyroid Stimulating Hormone (TSH) for
the Diagnosis of Congenital Hypothyroidism. J Trop Pediatr.
2008;54:74-7.
15. Rashmi, Seth A, Sekhri T, Agarwal A. Effect of
perinatal factors on cord blood thyroid stimulating hormone levels. J
Pediatr Endocrinol Metab. 2007;20:59-64.
16. Chan LY, Fok WY, Sahota D, Lau TK. Cord blood
thyroid-stimulating hormone level and risk of acidosis at birth.
European Journal of Obstet and Gyne and Reproductive Biol.
2006;124:173-7.
17. Chan LY, Leung TN, Lau TK. Influences of
perinatal factors on cord blood thyroid-stimulating hormone level. Acta
Obstetricia et Gynecologica Scandinavica. 2001;80:1014–8.
18. Lee MM, Moshang T Jr, Endocrine disorders of the
newborn. In: MacDonald, Mhairi G, Seshia, Mary MK, Mullett,
Martha D, ed. Avery’s Neonatology, 6th Edition; Philadelphia;
Lippincott Williams and Wilkins. 2005;1828–95.
19. Rico AJ, Prieto-Lloret J, Gonzalez C, Rigual R.
Hypoxia and acidosis increase the secretion of catecholamines in the
neonatal rat adrenal medulla: an in vitro study. Am J Physiol Cell
Physiol. 2005;289:C1417-C25.
20. Hugo. Stress, arousal and gene activation at
birth. News Physiol Sci. 1996;11:214–8.
21. Gülmezoglu AM, Mahomed K, Hofmeyr GJ, Nikodem VC,
Kramer T. Fetal and maternal catecholamine levels at delivery. J Perinat
Med. 1996;24:687-91.
22. Bistoletti P, Nylund L, Lagercrantz H, Hjemdahl
P, Ström H. Fetal scalp catecholamines during labor. Am J Obstet
Gynecol. 1983;147:785-8.
23. Desai M, Dabholkar C, Colaco MP. Thyroid function
in fullterm and preterm newborns. Indian J Pediatr. 1985;52:599-607.
24. Fletcher AJ, Gardner DS, Edwards CM, Fowden AL,
Giussani DA. Cardiovascular and endocrine responses to acute hypoxaemia
during and following dexamethasone infusion in the ovine fetus. J
Physiol. 2003;549(Pt. 1): 271-87.
25. Jellyman JK, Gardner DS, Edwards CM, Fowden AL,
Giussani DA. Fetal cardiovascular, metabolic and endocrine responses to
acute hypoxaemia during and following maternal treatment with
dexamethasone in sheep. J Physiol. 2005;567(Pt. 2):673-88.

