|
|
|
Indian Pediatr 2017;54: 1041-1046 |
 |
A New
Combined Vaccine Against Measles, Mumps, Rubella and Varicella
in India
|
|
* Nitin Shah,
#Raunak Parikh,
$Giacomo Casabona and
#Shafi Kolhapure
From *PD Hinduja National Hospital, and #GSK;
Mumbai, India; and $GSK, Wavre, Belgium.
Correspondence to: Dr Raunak Parikh, GSK, Dr Annie
Besant Rd, 400030 Mumbai, India.
Email: [email protected]
|
|
A quadrivalent MMRV (measles-mumps-rubella-varicella) combination
vaccine has recently been launched in India. This vaccine is highly
immunogenic, with seroconversion rates against all antigens reaching
96.6-100% at 42 to 56 days after the second vaccine dose in unvaccinated
children or in those previously vaccinated with MMR+/-V. Two doses
efficacy, against all varicella is 94.1% and effectiveness reaches 91%.
The most frequent solicited local adverse event after MMRV vaccine is
redness, and fever is the most common solicited general symptom. Higher
rates of fever and febrile convulsions compared to MMR+/-V have been
reported when used as first dose but not when used as the second of a
measles containing vaccine, irrespective of age of the second dose.
Keywords: Chicken pox, Measles-Mumps-Rubella
vaccine, Prevention, Varicella
|
|
M
easles, mumps, rubella and varicella are
vaccine-preventable diseases with the potential for significant
morbidity and even mortality [1-4]. Monovalent measles, mumps and
rubella vaccines were introduced in the 1960’s, with the first combined
measles-mumps-rubella (MMR) vaccine launched in the USA in 1971 [5].
Measles vaccine was introduced in Indian Universal Immunization
Programme (UIP) in 1985, and following the adoption of the global
measles and rubella strategic plan, it was recommended that
rubella-containing ‘MR’ vaccine should replace monovalent measles
vaccine for the first and second measles-containing vaccine (MCV) doses
[6]. However, the Indian Academy of Pediatrics (IAP) recommends a three
dose MMR schedule at 9 months, 15 months and 4-6 years [7].
Although first licensed in Germany in 1984, the
varicella vaccine is still not globally implemented in national
immunization programs. In India, the IAP recommends varicella dose 1 at
15 months followed by dose 2 between 4-6 years of age [7].
Alternatively, the second varicella dose can be administered below four
years of age provided that more than 3 months have elapsed since the
first dose [7]. Varicella
vaccination can be delivered either by monovalent varicella (V) or
quadrivalent measles-mumps-rubella-varicella (MMRV) vaccines [4]. There
are several potential advantages of using combination vaccines [8-11].
The number of vaccination visits and administrations (pricks) can be
reduced, thereby increasing vaccination acceptability, promoting
compliance and improving coverage rates [9-11]. Combination vaccines
simplify and shorten the reconstitution process and require less storage
space than single or low valency vaccines.
Although being globally available for over ten years,
MMRV has only recently been introduced in the Indian market [12]. This
review evaluates the clinical evidence for efficiency and safety of this
combination vaccine.
The Combined MMRV Vaccine
Priorix-Tetra (GSK, Belgium) was developed based on
the existing MMR (Priorix, GSK, Belgium) and varicella vaccines (Varilrix,
GSK, Belgium) [11]. Priorix-Tetra comprises a lyophilized powder
containing live, attenuated measles (Schwarz strain;
³103.0
CCID50), mumps (RIT 4385
strain; ³104.4
CCID50), rubella (Wistar RA
27/3 strain; ³103.0
CCID50) and varicella (Oka
strain; ³103.3
PFU) viruses [11]. It is indicated in children aged 1 to 12 years to
protect against measles, mumps, rubella and varicella [12]. In India,
the vaccine is reconstituted with the supplied diluent before
subcutaneous or intra-muscular administration [12].
Immunogenicity
Immunogenicity has been assessed in randomized
controlled multi-national trials involving over 3000 children (Web
Table I) [13-20]. Studies evaluated either two MMRV doses, or a
single MMRV dose after MMR
with/without varicella.
Two MMRV doses in the second year of life compared
with MMR+V followed by MMR
In the first, open, randomized controlled multicenter
study conducted in Germany, 970 healthy, MCV un-primed toddlers aged
10-21 months received either two MMRV doses, or separately administered
MMR+V followed by MMR; both doses were separated by 42 days [14]. At 42
days post-dose 2, seroconversion rates against all antigens were
³98.3% (MMRV group)
and ³97.5%
(MMR+V group). At this time point, the geometric mean titres (GMT) for
measles and mumps in the MMRV group were significantly higher than in
the MMR+V group. The rubella GMTs were not significantly different
between the groups, and as expected, the varicella GMTs were
significantly higher in MMRV recipients than in those who did not
receive a second varicella dose [14]. In another open, randomized
controlled multicenter study, from Austria and Germany, 494 healthy
toddlers aged 12-18 months received either two MMRV doses, or MMR+V
followed by MMR, both doses were separated by 42-56 days [15]. All
subjects in both groups seroconverted for measles, rubella and varicella
post-dose 2; 98.0% (MMRV) and 99.1% (MMR+V) seroconverted for mumps.
Measles and varicella GMTs were significantly higher in the MMRV group
[15]. Three years post-dose 2, antibodies against the vaccine antigens
persisted after both MMRV (³97.4%)
and MMR+V (³93.8%),
and all subjects remained seropositive to rubella [16].
MMRV as second dose of MMR vaccine
Healthy children aged 15-75 months primed with
single-dose MMR+V ³6
weeks previously, received a second dose of either MMRV or MMR+V in a
randomized open multicenter study in 390 children from Italy and Canada.
All MMRV subjects seroconverted against all four diseases (Web
Table I) [17]. Similar increases in GMTs from pre- to 42 days
post-dose 2 occurred in both groups for all antigens, but were
particularly marked for varicella. In another multicenter study, from
France, Germany and Italy, MMR-primed children received a second dose of
MMRV or MMR+V, either at 15 months–2 years or 2-6 years (Web Table
I) [18]. All subjects were subsequently vaccinated 42-56 days
later with a single dose of monovalent V. After the second dose of
either MCV, ³98.9%
subjects in both age groups had seroconverted against measles, mumps and
rubella. After one V dose, ³96.6%
subjects had seroconverted; this increased to 100% after the second V
dose [18].
Immunogenicity in Indian children
A phase III open randomized study undertaken at six
tertiary care centers in India administered either two doses of MMRV,
MMR followed by MMRV, or MMR followed by MMR+V (control) to children
aged 9-10 and 15 months [19]. After 43 days of MCV dose 2, 100%
seropositivity was recorded for MMR antigens in the MMRV/MMRV and
MMR/MMRV arms, and varicella in the MMRV/MMRV arm (Web Table I).
Seroconversion rates in the MMRV/MMRV and MMR/MMRV arms were
non-inferior to the controls. Multi-fold increases in GMTs were seen for
all antigens from 43 days post-dose 1 to 43 days post-dose 2,
particularly with respect to varicella [19].
Efficacy and effectiveness studies
Two doses of either MMRV or MMR (control), or
single-dose MMR followed by MMR+V (both doses separated by 42 days) were
compared in a large European multicenter study of 5803 children (aged
12-22 months; mean 14.2 months) [20]. After 3-years of follow-up,
varicella cases were confirmed in 37/2279 (MMRV), 243/2263 (MMR+V) and
201/743 (MMR) subjects, of which 2, 37 and 117, respectively were
moderate-to-severe. The efficacy of two-dose MMRV was 94.9% against all
varicella and 99.5% against moderate-to-severe varicella compared with
65.4% and 90.7%, respectively after MMR/MMR+V (i.e., single-dose V). The
authors concluded that two-dose varicella vaccine administered with a
short interval provided the optimum protection against the disease [20].
Efficacy until ³6
years post-vaccination has recently been demonstrated following two-dose
MMRV (95.0% (all varicella) and 99.0% (moderate-to-severe varicella),
compared with 66.9% and 90.2%, respectively after single-dose V) [21].
The effectiveness of MMRV and V vaccines was assessed
in 1084 children during seven varicella outbreaks in day-care centers in
Germany. The overall vaccine efficacy (71% in 352 eligible children,
P<0.001) was particularly sensitive to disease severity and number
of vaccine doses. The effectiveness of two-dose MMRV vaccine against any
varicella disease was 91% (P=0.001) [22].
Safety
Several reviews have concluded that MMRV and MMR+V
have comparable safety profiles [10,23]. A pooled analysis of three
large trials demonstrated that within four days of doses 1 and 2,
redness followed by swelling and pain were the most frequent solicited
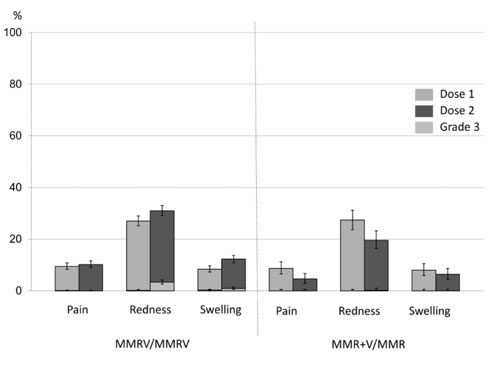
local symptoms after MMRV, MMR+V and MMR vaccines, (Fig. 1)
[11]. During the first 15 and 43 days post-vaccination, fever was the
most common solicited general symptom after all MCV doses [14,15];
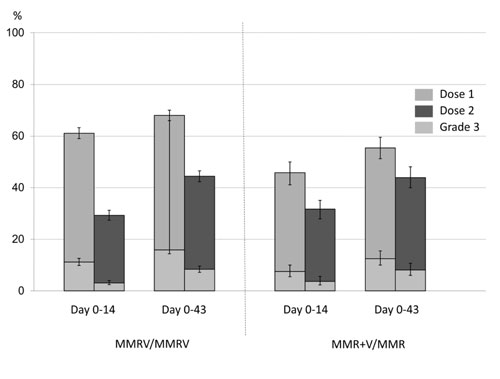
although, the incidence decreased after the second dose (Fig.
2) [11]. In the two largest European studies, rectal temperatures
³38.0°C were
more frequent after MMRV than MMR+V for 15 days post-dose 1 [(59.8%
vs 51.3%; P=0.023) [14] and (67.7% vs 48.8%; P<0.05)
[15]]. Unsolicited adverse events up to 43 days after vaccination did
not differ between the vaccine groups. In the largest trial, these were
reported in 45.4% and 43.3% after MMRV and MMR+V post-dose 1 and 41.1%
and 30.9%, respectively post-dose 2; these differences were not
statistically significant [14].
 |
|
Fig. 1 Pooled analysis of incidence
of solicited local adverse events within 4 days of
administration of two MCV doses. (created using data
provided in reference 11)
|
 |
|
Fig. 2 Pooled analysis of incidence
of fever within 14 and 43 days of administration of two MCV
doses. (created using data provided in reference 11)
|
‘Any’ rash occurs in 10.8-19.6% subjects during 43
days post-MMRV vaccination [10]. However, the incidence of measles,
rubella or varicella-like rashes, which are actively followed in
clinical trials, is generally low and not significantly different after
MMRV or MMR+V [11].
In the early trials, vaccine-related febrile
convulsions were recorded in one (out of 732) and two (out of 371)
subjects post-MMRV dose 1 [14,15]. In a German study of 226,267
MCV-unprimed children aged <5 years (90% aged between 11-23 months)
vaccinated with a first dose of MMR (n=111,241), MMR+V (n=32,370)
or MMRV (n=82,656), based on the "FC Jacobsen" definition, the
adjusted odds ratio (95% CI) for hospitalization with febrile convulsion
after the first MMRV dose (vs. MMR/MMR+V) during the main risk period
(5-12 days) was 2.4 [95% CI 1.5-4.0]. The risk of FC compared to
MMR/MMR+V was estimated as one additional seizure for every 2,747
vaccinated subjects [24].
Controlled trials have not indicated an increased
risk of fever after a second MMRV dose. The frequency of fever ( ³38.0°C)
post-dose 2 tends to be lower than post-dose 1 (Fig. 1 and
Table I); the incidence of fever after MMRV is also no
different than MMR+V [(36.8% vs 33.1%) [14]; (43.1% vs.
47.5%) [15]; Table
I]. Results of a post-licensure study in German children (MMR
98,348; MMR+V 10,315; MMRV 50,350 participants) suggest no statistically
significant increase of febrile convulsion after the second MMRV dose
(compared to MMR or MMR+V), even when administered in the second year
[25].
Table I Fever During 0-14 Days Following the Second Dose of MMR+/-V Vaccine in Controlled Clinical Trials
|
Reference |
Dosing schedule |
Vaccine administered |
Incidence of fever (%) |
|
|
as dose 2 |
|
|
|
|
|
|
N |
Any grade |
Grade 3 |
|
Schuster, et al. [14] |
10-21 mo age at 1st vaccination. |
MMRV |
725 |
36.8 |
3.6 |
|
2 doses MMRV 6 wks apart |
MMR+V |
236 |
33.1 |
4.7 |
|
Knuf, et al. [15] |
12-24 mo at age of 1st vaccination. |
MMRV |
356 |
43.1 |
6.0 |
|
2 doses MMRV 6-8 wks apart |
MMR+V |
116 |
47.5 |
5.7 |
|
Halperin, et al. [17] |
MMR + V ³6 wks before MMRV |
MMRV |
195 |
19.0 |
2.1 |
|
at 15-75 mo |
MMR+V |
195 |
16.0 |
3.1 |
|
Gillet, et al. [18] |
MMR ³6 wks before MMRV at 15-24 mo |
MMRV |
228* |
40.9 |
2.7 |
|
|
MMR+V |
230* |
31.5 |
2.8 |
|
MMR ³6 wks before MMRV at 2-6 y |
MMRV |
228# |
16.4 |
2.6 |
|
|
MMR+V |
230# |
20.7 |
2.6 |
|
Lalwani, et al. [19] |
MMR/MMRV or MMR/MMR+V at 9 and 15 mo |
MMRV |
159 |
13.2 |
1.3 |
|
|
MMR+V |
79 |
15.2 |
0 |
|
*Includes children aged 2-6 years; #Includes children aged
15-24 months. |
Safety in Indian children
In the phase III study in Indian children, pain was
the most common local symptom after the first vaccine dose (MMRV/MMRV
11.5%; MMR/MMRV 7.0%; MMR/MMR+V 10.7%), followed by redness (8.6%, 4.7%
and 3.6%, respectively) and swelling (4.6%, 2.9% and 3.6%, respectively)
[19]. Post-dose 2, solicited local symptoms occurred in <6.5% subjects
in any group. Any grade fever occurred in 13.2-32.2% children during the
first 15 days post-vaccination, and there was no significant difference
between the incidences of fever after the different vaccines
(overlapping confidence intervals). The incidence of fever was lower
post-dose 2 (Table I). Post-dose 1,
³1 unsolicited
adverse event occurred in 20.6% (MMRV/MMRV), 21.7% (MMR/MMRV) and 20.0%
subjects (MMR/MMR+V), compared with 10.6%, 10.0% and 12.2%, respectively
post-dose 2. The most frequent unsolicited adverse events included upper
respiratory tract infection, cough, nasopharyngitis and rhinitis. No
episode of febrile convulsion occurred during six weeks
post-vaccination. However, this was a relatively small trial and may not
necessarily have captured a rare side effect, which can only be seen
post-marketing when more doses are administered.
Dosing Schedule
MMRV vaccines have been used for over a decade
worldwide and considerable post-marketing experience exists. In USA [26]
and Germany [27], where the first varicella dose coincides with first
MCV dose, MMR+V is preferred over MMRV due to higher rates of fever and
two-fold higher febrile convulsion rates. However, there is no increased
risk of febrile convulsion when MMRV vaccine is administered as 2nd dose
of MCV, irrespective of the age at which the second dose is
administered.
In USA, where the recommended age for the second
routine MMRV dose is 4-6 years, the second dose may be administered
three months after first dose. In this situation, MMRV vaccine is
preferred (over MMR+V) in children ³15
months to 12 years of age [26]. In Germany, MMRV can be administered as
a second dose in 15- to 23-month-old children [27]; and in Australia
[28], MMRV is exclusively provided to children at 18 months, following
previous vaccination with MMR at 12 months.
In India, first dose of MCV is administered at 9-12
months; children receive their first varicella dose with 2nd dose of MCV
in the second year of life. Considering the benefit-risk profile of
MMRV, there are many possible scenarios for vaccine implementation in
India. Complexity is added by individual practitioner-preference related
to timing and number of doses of MCV. Two MMRV doses can be administered
six weeks apart in children ³12
months, but taking into account the current IAP recommendations for MMR
and Varicella vaccines, and the individual preferences of private
practitioners, the most feasible likely schedules are suggested in
Table II. Additionally, for catch-up MMRV vaccination up to
12 years, as well as for children who miss MCV at 9-12 months, two MMRV
doses can be administered with an interval of
³6 weeks.
Table II Feasible Usage Schedules for MMRV Vaccine in India
|
Age |
|
9 mo |
15 mo |
18 mo |
4-6 yr |
|
Option 1 |
MCV1 |
MMRV |
- |
MMRV |
|
Option 2 |
MCV1 |
MMRV |
V |
MMR |
|
Option 3 |
MCV1 |
MMRV |
MMRV |
- |
|
MCV1: MMR, M or MR. M: monovalent measles vaccine; V:
monovalent varicella vaccine; MR: bivalent measles and rubella
vaccine; MMR: trivalent measles, mumps and rubella vaccine;
MMRV: quadrivalent measles, mumps, rubella and varicella
vaccine. |
Co-administration
The number of clinic appointments can be further
reduced, and the acceptability of immunization schedules improved, by
co-administering MMRV with vaccines against other diseases at the same
visit. MMRV has been administered concurrently with the childhood
vaccines that are scheduled at similar times; for example,
diphtheria-tetanus-acellular pertussis-inactivated polio-virus
vaccine-hepatitis B/Haemophilus influenzae (DTPa-HBV-IPV/Hib), hepatitis
B, meningococcal and pneumococcal vaccines [29-32].
Concurrent MMRV plus DTPa-HBV-IPV/Hib vaccines, were
compared with single doses of either vaccine in 451 healthy children
aged 12-23 months. At 42 days post-vaccination, no differences in
seroconversion rates for measles, mumps, rubella and varicella, or
immune responses to the DTPa-HBV-IPV/Hib antigens were seen. There were
no differences in GMTs between the two groups, apart from measles, which
were higher after MMRV than MMR+V. Additionally, when co-administered,
there was no evidence of any exacerbation in the reactogenicity of
either vaccine [29]. A large multi-center trial comparing either
concurrent MMRV, Hib/HepB and DTaP, or MMRV or MMR+V followed by Hib/HepB
and DTaP 42 days later, in 1915 healthy children aged 12-15 months
showed similar safety profiles and antibody responses to all antigens in
all groups [30].
Conclusion
MMR and varicella vaccines have been available in the
market in India for several years and quadrivalent MMRV, which
simultaneously protects against all four viruses, has recently become
available. Fever rates are usually high when MMRV vaccine is
administered as first dose of MCV; fever (and febrile convulsion) rates
are higher than for MMR or concomitant MMR+V. However, controlled trials
indicate comparable fever rates after a second MMRV dose or when MMRV
dose is administered as 2nd dose of MCV.
In India, as MMRV is indicated for children
³12 months, this
vaccine will predominantly be administered as 2nd MCV. One or two MMRV
doses can therefore be introduced into the Indian immunization schedule
to offer broad disease protection and to provide the general advantages
of combination vaccines to vaccinees, parents and physicians.
Acknowledgements: GSK advisory board on the MMRV
vaccine (Nitin Shah, Raju Shah, Gaurav Gupta, Aniruddha Chatterjee,
Monjori Mitra). Julia Donnelly (freelance for XPE Pharma, Wavre, Belgium
C/O GSK) for medical writing assistance and Adrian Kremer (employed by
XPE Pharma, Wavre, Belgium, C/O GSK) for publication coordination.
Contributors: NS, RP, GC and SK contributed to
literature review, interpretation of the data, and critical review of
the paper for important intellectual content.
Funding: GlaxoSmithKline Biologicals SA.
Competing interests: RP, GC and SK are
employees of the GSK group of companies and GC holds shares in the GSK
group of companies as part of his employee remuneration. NS declares
receipt of consultancy fees from GSK, Pfizer, Sanofi Pasteur, MSD,
Abbott Vaccines, Novo Medisciences, Bharat Biotech, Cipla India and
Wockhardt vaccines.
References
1. World Health Organisation. Mumps Virus Vaccines:
WHO Position Paper. Wkly Epidemiol Rec. 2007;82:51-60.
2. World Health Organisation. Measles Vaccines: WHO
Position Paper. Wkly Epidemiol Rec. 2009;84:349-60.
3. World Health Organisation. Rubella Vaccines: WHO
Position Paper. Wkly Epidemiol Rec. 2011;86:301-16.
4. World Health Organisation. Varicella and Herpes
zoster Vaccines: WHO Position Paper. Wkly Epidemiol Rec. 2014;89:265-87.
5. Historic Dates and Events Related to Vaccines and
Immunization. Available from: http://www.immunize.org/timeline/.
Accessed July 18, 2016.
6. World Health Organization. Global Measles and
Rubella Strategic Plan, 2012–2020. Geneva; WHO 2012. Available from:http://www.who.int/immunization/documents/control/ISBN_978_92_4_150339_6.
Accessed July 18, 2016.
7. IAP Immunization Timetable 2016. Available from:
http://www.iapindia.org/page.php?id=129. Accessed April 13, 2017.
8. Mathew JL. Evidence-based options to improve
routine immunization. Indian Pediatr. 2009;46:993-6.
9. Marshall GS, Happe LE, Lunacsek OE, Szymanski MD,
Woods CR, Zahn M, et al. Use of combination vaccines is
associated with improved coverage rates. Pediatr Infect Dis J.
2007;26:496-500.
10. Ma SJ, Li X, Xiong YQ, Yao AL, Chen Q.
Combination measles-mumps-rubella-varicella vaccine in healthy children:
A systematic review and meta-analysis of immunogenicity and safety.
Medicine. 2015;94:e1721.
11. Czajka H, Schuster V, Zepp F, Esposito S, Douha
M, Willems P. A combined measles, mumps, rubella and varicella vaccine (Priorix-Tetra):
Immunogenicity and safety profile. Vaccine. 2009;27:6504-11.
12. Priorix Tetra Full Prescribing Information
Version PRT/PI/IN/2017/02. Available from: GlaxoSmithKline
Pharmaceuticals Ltd Dr. Annie Besant Road, Worli, Mumbai-400030 (India).
13. Knuf M, Zepp F, Meyer CU, Habermehl P, Maurer L,
Burow HM, et al. Safety, immunogenicity and immediate pain of
intramuscular versus subcutaneous administration of a
measles-mumps-rubella-varicella vaccine to children aged 11-21 months.
Eur J Pediatr. 2010;169:925-33.
14. Schuster V, Otto W, Maurer L, Tcherepnine P,
Pfletschinger U, Kindler K, et al. Immunogenicity and safety
assessments after one and two doses of a refrigerator-stable tetravalent
measles-mumps-rubella-varicella vaccine in healthy children during the
second year of life. Pediatr Infect Dis J. 2008;27:724-30.
15. Knuf M, Habermehl P, Zepp F, Mannhardt W, Kuttnig
M, Muttonen P, et al. Immunogenicity and safety of two doses of
tetravalent measles-mumps-rubella-varicella vaccine in healthy children.
Pediatr Infect Dis J. 2006;25:12-8.
16. Knuf M, Zepp F, Helm K, Maurer H, Prieler A,
Kieninger-Baum D, et al. Antibody persistence for 3 years
following two doses of tetravalent measles-mumps-rubella-varicella
vaccine in healthy children. Eur J Pediatr. 2012;171:463-70.
17. Halperin SA, Ferrera G, Scheifele D, Predy G,
Stella G, Cuccia M, et al. Safety and immunogenicity of a
measles-mumps-rubella-varicella vaccine given as a second dose in
children up to six years of age. Vaccine. 2009;27:2701-6.
18. Gillet Y, Steri GC, Behre U, Arsene JP, Lanse X,
Helm K, et al. Immunogenicity and safety of
measles-mumps-rubella-varicella (MMRV) vaccine followed by one dose of
varicella vaccine in children aged 15 months-2 years or 2-6 years primed
with measles-mumps-rubella (MMR) vaccine. Vaccine. 2009;27:446-53.
19. Lalwani S, Chatterjee S, Balasubramanian S,
Bavdekar A, Mehta S, Datta S, et al. Immunogenicity and safety of
early vaccination with two doses of a combined measles-mumps-rubella-varicella
vaccine in healthy Indian children from 9 months of age: a phase III,
randomised, non-inferiority trial. BMJ Open. 2015;5:e007202.
20. Prymula R, Bergsaker MR, Esposito S, Gothefors L,
Man S, Snegova N, et al. Protection against varicella with two
doses of combined measles-mumps-rubella-varicella vaccine versus one
dose of monovalent varicella vaccine: a multicentre, observer-blind,
randomised, controlled trial. Lancet. 2014;383:1313-24.
21. Czajka H, Brzostek J, Gabutti G, Gasparini R,
Desole MG, Pazdiora P, et al. Comparison of six year efficacy
between one or two doses of live varicella virus-containing vaccines: a
randomized, placebo-controlled trial. Presented at European Society for
Paediatric Infectious Diseases - 33rd Annual Meeting. 12-16 May 2015;
Leipzig Germany: ESPID-0644.
22. Spackova M, Wiese-Posselt M, Dehnert M,
Matysiak-Klose D, Heininger U, Siedler A. Comparative varicella vaccine
effectiveness during outbreaks in day-care centres. Vaccine.
2010;28:686-91.
23. Dhillon S, Curran MP. Live attenuated measles,
mumps, rubella, and varicella zoster virus vaccine (Priorix-Tetra).
Paediatr Drugs. 2008;10:337-47.
24. Schink T, Holstiege J, Kowalzik F, Zepp F, Garbe
E. Risk of febrile convulsions after MMRV vaccination in comparison to
MMR or MMR+V vaccination. Vaccine. 2014;32:645-50.
25. Hense S, Schink T, Garbe E. 2nd immunization
against measles, mumps, rubella with MMRV, MMR or MMR+V in Germany and
risk of febrile convulsions. Presented at International Society for
Pharmacoepidemiology - 29th International Conference. 25-28 August 2013;
Montreal, Canada: ID 24.
26. Marin M, Broder KR, Temte JL, Snider DE, Seward
JF. Use of combination measles, mumps, rubella, and varicella vaccine:
recommendations of the Advisory Committee on Immunization Practices
(ACIP). MMWR. Recommen-dations and Reports. 2010;59:1-12.
27. Recommendations of the Standing Committee on
Vaccination (STIKO) at the Robert Koch Institute – 2016/2017.
Epidemiologisches Bulletin. 2016;34:301-38.
28. Measles, Mumps, Rubella, Varicella Vaccine -
Information for health professionals. Available from:
http://www.immunise.health.gov.au/internet/immunise/publishing.nsf/Content/IT0167-cnt/$File/final-mmrv-provider-booklet.pdf.
Accessed April 21, 2017.
29. Zepp F, Behre U, Kindler K, Laakmann KH,
Pankow-Culot H, Mannhardt-Laakmann W, et al. Immunogenicity and
safety of a tetravalent measles-mumps-rubella-varicella vaccine
co-administered with a booster dose of a combined diphtheria-tetanus-acellular
pertussis-hepatitis B-inactivated poliovirus-haemophilus influenzae type
b conjugate vaccine in healthy children aged 12-23 months. Eur J Pediatr.
2007;166:857-64.
30. Shinefield H, Black S, Thear M, Coury D,
Reisinger K, Rothstein E, et al. Safety and immunogenicity of a
measles, mumps, rubella and varicella vaccine given with combined
Haemophilus influenzae type b conjugate/hepatitis B vaccines and
combined diphtheria-tetanus-acellular pertussis vaccines. Pediatr Infect
Dis J. 2006;25:287-92.
31. Vesikari T, Karvonen A, Lindblad N, Korhonen T,
Lommel P, Willems P, et al. Safety and immunogenicity of a
booster dose of the 10-valent pneumococcal nontypeable Haemophilus
influenzae protein D conjugate vaccine coadministered with
measles-mumps-rubella-varicella vaccine in children aged 12 to 16
months. Pediatr Infect Dis J. 2010;29:e47-56.
32. Vesikari T, Karvonen A, Bianco V, Van der Wielen
M, Miller J. Tetravalent meningococcal serogroups A, C, W-135 and Y
conjugate vaccine is well tolerated and immunogenic when co-administered
with measles-mumps-rubella-varicella vaccine during the second year of
life: An open, randomized controlled trial. Vaccine. 2011;29: 4274-84.
|
|
|
 |
|

