|
|
|
Indian Pediatr 2017;54:749-751 |
 |
Yield of Imaging
Performed as per Indian Society of Pediatric Nephrology
Guidelines in Children with Urinary Tract Infection
|
|
*# Rajiv Sinha ,
*Devdeep
Mukherjee , *#Jayati
Sengupta, #$Subhasis
Saha and *$Sushmita
Banerjee
From Departments of Pediatric Nephrology; *Institute
of Child Health, #AMRI Hospitals and $Calcutta
Medical Research Institute, Kolkata; India
Correspondence to: Dr Sushmita Banerjee, Pediatric
Nephrologist, Calcutta Medical Research Institute, Kolkata, India.
Email: [email protected]
Received: May 04, 2016;
Initial review: December 13, 2016;
Accepted: June 29, 2017.
|
Objectives: To assess yield of imaging performed
as per Indian Society of Pediatric Nephrology (ISPN) urinary tract
infection (UTI) guideline. Methods: Ultrasonography (USG),
voiding cystourethrography (VCUG) and dimercaptosuccinic-acid (DMSA)
scintigraphy were performed in 183 children (age 0-5y) with first
episode (age 0-1y) of UTI or recurrent (age <5y) UTI, as per ISPN
recommendations.Results: Significant abnormalities were detected
in 110 (63%), with vesicoureteric reflux (VUR) grades 3-5 in 31% and
renal scars in 43%. Combined USG and DMSA had a negative predictive
value of 94% for significant VUR. Conclusion: ISPN guideline
resulted in a high yield of detection of significant abnormalities.
Keywords: Diagnosis, Evaluation, Management, Vesicoureteric
reflux.
|
|
G
uidelines for imaging after urinary tract
infection (UTI) in young children vary in different countries [1-4]. The
Indian Society of Pediatric Nephrology (ISPN) recommends that infants
after first UTI, and children between 1 to 5 years with recurrent UTI
should be investigated with ultrasonography (USG), voiding
cystourethrography (VCUG) and dimercaptosuccinic acid (DMSA)
scintigraphy [1]. We prospectively analyzed case records of children
with UTI, who had all these investigations performed as per ISPN
guidelines to assess the frequency of significant renal abnormalities in
such children.
Methods
We enrolled children with diagnosis of UTI between
September 2013 and August 2015 at two tertiary care centers in Kolkata,
India. UTI was diagnosed as per ISPN guideline [1] i.e. positive
urine culture in a child having symptoms suggestive of UTI. Urine was
collected by clean catch, supra-pubic aspiration or urethral
catheterization. We included all children with first episode of UTI in
infancy, and recurrent ( ³2)
UTI up to 5 years of age. Clinical symptoms were recorded. Symptoms of
constipation, poor urinary stream, hesitancy, straining or dribbling
were classified as bowel bladder dysfunction (BBD). USG was performed
early after diagnosis, MCUG after confirming UTI resolution, and DMSA
after a minimum of 2 months. Significant abnormalities were defined as:
hydronephrosis with renal pelvic diameter
³10 mm, ureteric
dilatation, structural bladder abnormalities (thickened wall or
diverticulum) and/or bladder residue >20 mL on USG; vesico-ureteric
reflux (VUR) grades 3 to 5, bladder or urethral abnormalities on VCUG;
or parenchymal scars on DMSA.
Results
We enrolled 183 consecutive children. Seven were
excluded as they did not complete all three investigations. Out of 176
enrolled children (90 girls), 93 (52.8%) were infants. Among infants,
56% had recurrent UTI, 84% febrile UTI and 13% had BBD. Among 83
children aged between one and five years, 89% were febrile and 24% had
BBD.
Significant abnormalities were present in 110 (63%)
patients: 61 among infants and 49 in 1-5 year-olds. Sixty (54%) were
girls, 105 (95%) had febrile UTIs and 32 (29%) had symptoms of BBD (Table
I). The common abnormalities detected were: hydronephrosis
(unilateral in 47, bilateral in 12), renal scars (unilateral in 52,
bilateral in 24), and VUR grades 3-5 (unilateral in 29, bilateral in
25). Thirty children had post-void bladder residue of >20 mL, three had
posterior urethral valves and another three had bladder trabeculations.
Table 1 Comparison of Patients With and Without Significant Abnormalities on Imaging
|
Characteristics |
Infants n=93 |
Children (aged 1-5y) n=83 |
|
NSA, n=32 |
SA, n=61 |
NSA, n=34 |
SA, n=49 |
|
Male gender |
16 (50) |
35 (57.4) |
20 (58.8) |
15 (30.6)* |
|
Febrile |
18 (56.3) |
60 (98.4)* |
29 (85.3) |
45 (91.8) |
|
Symptoms of BBD |
0 |
12 (19.7)* |
0 |
20 (40.8)* |
|
Recurrent UTI |
13 (40.6) |
39 (63.4)* |
NA |
NA |
|
*P<0.05, SA= significant abnormality, NSA: no significant
abnormality, BBD: bladder bowel dysfunction; Values in No.(%). |
Significant abnormalities were more common in
patients with febrile UTI (P=0.001) or symptoms of BBD (P<0.001).
Significant abnormalities were more common in infants who had febrile (P<0.001)
or recurrent UTI (P=0.047), in girls aged 1-5 yr (P=0.013),
and in patients with BBD (P<0.001 and P=0.007) (Table
I).
Thirty (17%) patients had abnormal DMSA and/or
abnormal VCUG in presence of a normal USG (Fig. 1a). The
two investigations that in combination would have detected the most
number of significant abnormalities were USG and DMSA. These, if
performed without VCUG, would have missed significant VUR in only 4 (2%)
patients. Of these 4, all were infants with febrile UTIs, 3 were males
with unilateral grade 3 VUR while one girl had unilateral grade 4 VUR.
The negative predictive value of a normal USG + DMSA for excluding VCUG
abnormality was 94%.
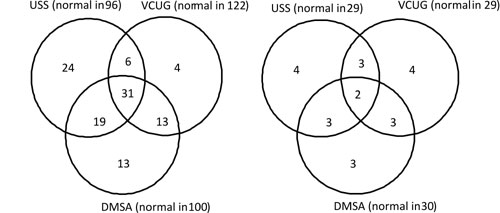 |
| (a) |
(b) |
|
Fig. 1 Venn diagrams showing results
of imaging in the whole study population (a) and in infants with
first UTI (b) (The figures within the circles represent numbers
with significant abnormalities).
|
In infants, USG (P=0.012) and DMSA (P=0.033)
abnormalities but not VCUG (P=0.088) abnormality was
significantly more common in those with recurrent UTI. 29 infants with a
first UTI had normal USG of which 4 had significant VUR, 3 had renal
parenchymal scars and 3 had both VUR and scars (Fig. 1b).
Significant abnormalities would have been missed in 10/41 (25%) infants
with first episode of UTI if NICE (National Institute for Health and
Clinical Excellence, UK) [2] or AAP (American Academy of Pediatrics) [3]
guidelines were followed, which do not advocate further investigation if
USG is normal, and there are no atypical features or risk factors.
Discussion
In children, UTI may unmask underlying
structural or functional anomalies of the urinary tract, and may be
associated with renal parenchymal scars. Debate persists on the range of
follow-up investigations [5-7], and is reflected in the differences in
published guidelines [1-4]. Our assessment of the yield of
investigations performed as per ISPN guidelines [1], focusing on the
highest risk groups, revealed 63% children to have significant
abnormalities. Our study also identified febrile UTI and symptoms of BBD
as groups particularly needing close attention and follow-up.
While all guidelines are in agreement of more
extensive investigation for children with recurrent UTI, prime
differences lie in the imaging of infants after first UTI. The ISPN [1]
advocates all three investigations (USG, VCUG and DMSA), in this group.
NICE and AAP guidelines [2,3] are more selective in their use of
VCUG/DMSA and rely on the result of USG. Similar to observations by Tse,
et al. [8], we demonstrated that had the latter guidelines been
followed, 25% of significant abnormalities would have been missed among
infants with UTI.
Previous publications have suggested that a normal
DMSA may obviate the need for VCUG and vice-versa [9-14]. In our
study, if only DMSA was done, we would have missed significant VUR in 10
patients (Fig. 1a). Performing only VCUG would have missed
renal scars in 32 patients. Similar to a recent publication by Lee,
et al. [15], we found that the combination of normal USG and DMSA
had a high negative predictive value of 94% for VUR.
Our results should be interpreted with caution
because of the relatively small sample size. Also, the study was
performed in tertiary centers where there is the possibility of a
referral bias.
In conclusion, the current study demonstrates that
compliance to ISPN UTI guidelines for infants with first UTI and
children below 5 years with recurrent UTI, results in a high yield of
detection of significant abnormalities. The ISPN guidelines seem to be
the most appropriate in the Indian scenario, where early specific
diagnosis alerts caregivers to the need for requisite follow-up.
Acknowledgement: Dr Surupa Basu (Department of
Biochemistry, Institute of Child Health) and Mrs Sayantani Majumdar
(Statistician) provided valuable inputs with statistical analysis.
Contributors: All authors contributed to data
collection. SS performed the VCUG. RS, DM and SB were involved in data
analysis and writing of the manuscript. JS and SS reviewed and edited
the paper.
Funding: None; Competing interests:
None stated.
|
What This Study Adds?
• In Indian children (age 0-5 y) with UTI, following the ISPN
guidelines for imaging resulted in a high yield of detection of
significant underlying abnormalities.
|
References
1. Indian Society of Pediatric Nephrology;
Vijayakumar M, Kanitkar M, Nammalwar BR, Bagga A. Revised statement on
management of urinary tract infections. Indian Pediatr. 2011;48:709-17.
2. National Institute for Health and Clinical
Excellence. Urinary Tract Infection in Children: Diagnosis, Treatment
and Long Term Management. 2007. Clinical Guidelines, No. 54.
3. Subcommittee on Urinary Tract Infection, Steering
Committee on Quality Improvement and Management, Roberts KB. Urinary
tract infection: clinical practice guideline for the diagnosis and
management of the initial UTI in febrile infants and children 2 to 24
months. Pediatrics. 2011;128:595-610.
4. Ammenti A1, Cataldi L, Chimenz R, Fanos V, La
Manna A, Marra G, et al. Febrile urinary tract infections in
young children: Recommendations for the diagnosis, treatment and
follow-up. Acta Paediatr. 2012;101:451-7.
5. South M. Radiological investigations following
urinary tract infection: changes in Australian practice. Arch Dis Child.
2009;94:927-30.
6. Tullus K. Outcome of post-infectious renal
scarring. Pediatr Nephrol. 2015;30:1375-7.
7. McDonald K, Kenney I. Paediatric urinary tract
infections: A retrospective application of the National Institute of
Clinical Excellence guidelines to a large general practitioner referred
historical cohort. Pediatr Radiol. 2014;44:1085-92.
8. Tse NK, Yuen SL, Chiu MC, Lai WM, Tong PC. Imaging
studies for first urinary tract infection in infants less than 6 months
old: can they be more selective? Pediatr Nephrol. 2009;24:1699-703 .
9. Leonardo CR, Filgueiras MF, Vasconcelos MM,
Vasconcelos R, Marino VP, Pires C, et al. Risk factors for renal
scarring in children and adolescents with lower urinary tract
dysfunction. Pediatr Nephrol. 2007;22: 1891-6.
10. Ajdinoviæ B, Jaukoviæ L, Krstiæ Z, Dopuda M.
Technetium-99m-dimercaptosuccinic acid renal scintigraphy in children
with urinary tract infections. Hell J Nucl Med. 2006;9:27-30.
11. Nammalwar BR, Vijayakumar M, Sankar J, Ramnath B,
Prahlad N. Evaluation of the use of DMSA in culture positive UTI and
culture negative acute pyelonephritis. Indian Pediatr. 2005;42:691-6.
12. Hansson S, Dhamey M, Sigström O, Sixt R, Stokland
E, Wennerström M, et al. Dimercapto-succinic acid scintigraphy
instead of voiding cystourethrography for infants with urinary tract
infection. J Urol. 2004;172: 1071-3.
13. Tseng NH, Lin WJ, Lo WT, Wang SR, Chu ML, Wang
CC. Does a normal DMSA obviate the performance of voiding
cystoureterography in evaluation of young children after their first
urinary tract infection? J Pediatr. 2007;150:96-9.
14. Preda I, Jodal U, Sixt R, Stokland E, Hansson S.
Normal dimercaptosuccinic acid scintigraphy makes voiding
cystourethrography unnecessary after urinary tract infection. J Pediatr.
2007;151:581-4.
15. Lee HY, Soh BH, Hong CH, Kim MJ, Han SW. The
efficacy of ultrasound and dimercaptosuccinic acid scan in predicting
vesicoureteral reflux in children below the age of 2 years with their
first febrile urinary tract infection. Pediatr Nephrol. 2009;24:2009-13.
|
|
|
 |
|

