Subgaleal hematoma (SGH) is a potentially
life-threatening extra-cranial bleed that occurs most commonly in
neonates after difficult instrumental deliveries(1). It can present
acutely as hemorrhagic shock or anemia, and later as neonatal
hyperbili-rubinemia(1,2). We report a case of SGH resulting in anemia
and severe hyper-bilirubinemia requiring an unusually large number of
exchange transfusions.
Case Report
Baby M, a 3000 g male infant, was delivered at 38
weeks gestation by emergency cesarean section for non-progress of labor.
There was no evidence of perinatal asphyxia or encephalopathy. On day 2
he was referred to our hospital for pallor and a scalp swelling. At
admission, his hemodynamic and respiratory parameters were stable, his
occipito-frontal circumference (OFC) was 37 cm, there was severe pallor
and a SGH extending from the upper eyelids to the nape of the neck.
There was no bruising or bleeding from any site.
His hemoglobin was 6.9 g/dL, platelets were adequate
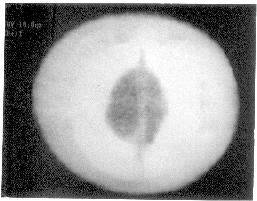
and there was no evidence of coagulopathy. Computed tomographic (CT)
scan of the head revealed circumferential scalp swelling crossing suture
boundaries suggest-ing of SGH. The intracranial structures were normal (Fig
. 1).
 |
|
Fig.1. NCCT scan head showing circumferential
scalp swelling (patchy hypoattenuation) around skull bones (dense
white). |
He was treated with vitamin K injection and blood
transfusions (15 ml/Kg) twice on the day of admission. On day 5 of life,
he developed jaundice with total serum bilirubin (TSB) of 16 mg/dL and
phototherapy was started. The blood groups of the baby and mother were
both B positive. The direct Coombs test was negative and
glucose-6-Phosphate dehydrogenase level was normal. The reticulocyte
count was 2%. Inspite of phototherapy, the TSB progressively rose and
hence he underwent a double volume exchange transfusion (DVET) on day 6.
He subsequently underwent 3 more DVET’s within next 36 hrs, all for TSB
values above 20 mg/dL. On the 10th day the TSB (predominantly
unconjugated) again rose rapidly and hence he underwent 3 more DVET’s
over the next 2 days. All donor blood aliquots were of B positive group,
and none was more than 5 days old. There was no evidence, either
clinically or hematologically, of a hemolytic reaction after any of the
DVETs. It is our policy to send a sample of blood from all blood
transfusion bags for G6PD assay, and we found in this case that none of
the donors were G6PD deficient. The patient received highly intensive
single surface phototherapy with special blue lights (Philips TL 52 with
irradiance of 18 W/cm2/nm) throughout the duration of hyper-bilirubinemia.
We did not drain or attempt to aspirate the SGH. No medication was used
to treat the hyperbilirubinemia. At discharge he was mildly jaundiced,
neurologically normal with OFC 33.5 cm. At follow up after one month the
size of the swelling had decreased and brain stem evoktesponse
audiometry was normal.
Discussion
This case of massive SGH is unusual for the large
number of exchange transfusions required for treating the
hyperbilirubinemia. The sub-aponeurotic space accommodates as much as
250 ml of blood when filled to just 1 cm in thickness(3). It is
potentially life threatening because of the potential of hemorrhagic
shock.
The reported incidence of SGH ranges from 1.6-3/1000
live births(2,4). The risk factors are instrumental delivery, prolonged
second stage of labor; precipitate labor, coagulopathy, prematurity,
large infants, fetal dystocia and severe head moulding(2,4-7). The
incidence of SGH in ventouse-associated deliveries is 60 times higher
than other modes of delivery(5). Male preponderance was reported(5). The
index patient was a male child delivered after labour of 48 hours. There
was no instrumentation or bleeding diathesis.
Subgaleal hematoma is associated with hypoxic
ischemic encephalopathy, head trauma such as intracranial hemorrhage,
skull fracture and cerebral edema in various studies(8). In a study by
Govaret et al.(9), 14 of 27 babies with SGH revealed various
abnormalities of parietal bones on CT scan. Three mechanisms of bleeding
were suggested: linear skull fracture, suture diastases and
fragmentation of the superior margin of parietal bone. In our patient
there was no skull fracture, sutural diastases or intracranial bleed on
cranial CT scan.
The clinical presentation varies from insidious onset
of anemia to hypovolemic shock(1,2). In a study of 101 subjects over a
period of 30 months, the clinical presentation of SGH was
hyperbilirubinemia in 57 patients (56.4%), anemia requiring blood
transfusion in 32.7% and shock in 3 babies. Four patients developed
severe unconjugated hyperbili- rubinemia, which required exchange
trans-fusions(2). Our patient presented with anemia requiring blood
transfusions at admission and later developed severe hyperbilirubinemia
requiring seven DVET’s. To the best of our knowledge this is the maximum
number of DVET’s a patient with SGH has undergone so far in published
medical literature.
Phototherapy appears to have been ineffective in our
patient, despite adequate light intensity and wavelength. This is
somewhat contrary to the experience of Tan et al.(10) who found
phototherapy to be equally effective in the cephalhematoma. The patient
was started on prophylactic antibiotics after the third exchange, and
did not develop sepsis during the hospital stay.
An important issue is whether we should have
aspirated the blood at the outset to prevent repeated DVETs. The
literature is ambiguous about this. There is a sizeable risk of
introducing infection during aspiration of an otherwise sterile hematoma,
and hence aspiration has generally been recommended only when one wishes
to exclude the presence of an infected cephalhematoma(11).
The literature is also silent about the possible use
of glucoronyl transferase inducers such as phenobarbitone, or the use of
heme oxygenase inhibitors, such as metalloporphyrins, in the presence of
large hematomas to prevent the occurrence of hyperbilirubinemia. One
animal study has shown that single dose tin protoporphyrin does not
reduce bilirubin formation following artificially created hematomas(12).
The mortality rate following SGH is 14-17%(4,5). The
most important risk factors associated with death following SGH include
severe anemia requiring urgent blood transfusion within first 12 hours
of birth and significant birth asphyxia(5). After the acute phase, SGH
resolves over 2 to 3 weeks. Newborns with isolated SGH who survive the
acute episode show no evidence of subsequent long-term major
neurological deficit or developmental delay(4,8).
Contributors: SD managed the patient and
co-drafted the manuscript, AS drafted the manuscript, AN supervised the
management of the patient and the drafting of the manuscript.
Funding: None.
Competing interests: None stated.
